The Cover Story
Vol.71,No.3 Cover
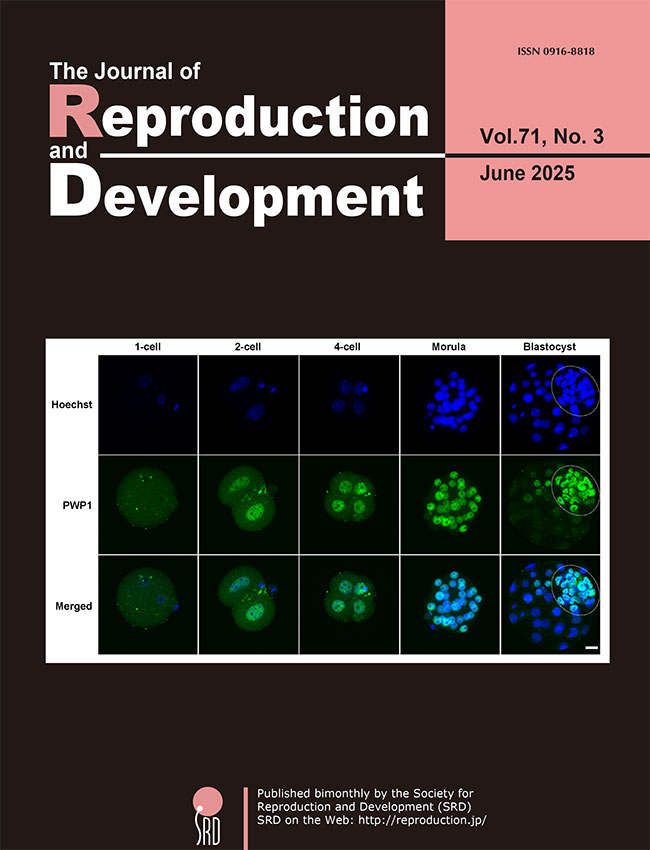
After fertilization, dramatic changes in epigenetic regulations and zygotic genome activation (ZGA) occur, eventually leading to a transition from totipotency to pluripotency. However, the regulation of these processes during preimplantation development remains unclear. Yamamoto et al. investigated the functional roles of periodic tryptophan protein 1 (PWP1) in mouse preimplantation embryos (Yamamoto
et al. Pwp1 inhibition impairs the development and early lineage commitment of mouse preimplantation embryos, pp. 168–174). The expression of
Pwp1 increased during ZGA, and the PWP1 protein was predominantly localized in the nuclei of the inner cell mass at the blastocyst stage (cover photo).
Pwp1 knockdown reduced the developmental potential of mouse preimplantation embryos, accompanied by prolonged expression of the ZGA-related genes at the morula stage and altered expression of cell lineage-related genes at the blastocyst stage. These findings suggest that PWP1 is essential for the regulation of early embryonic development.
Vol.71,No.2 Cover
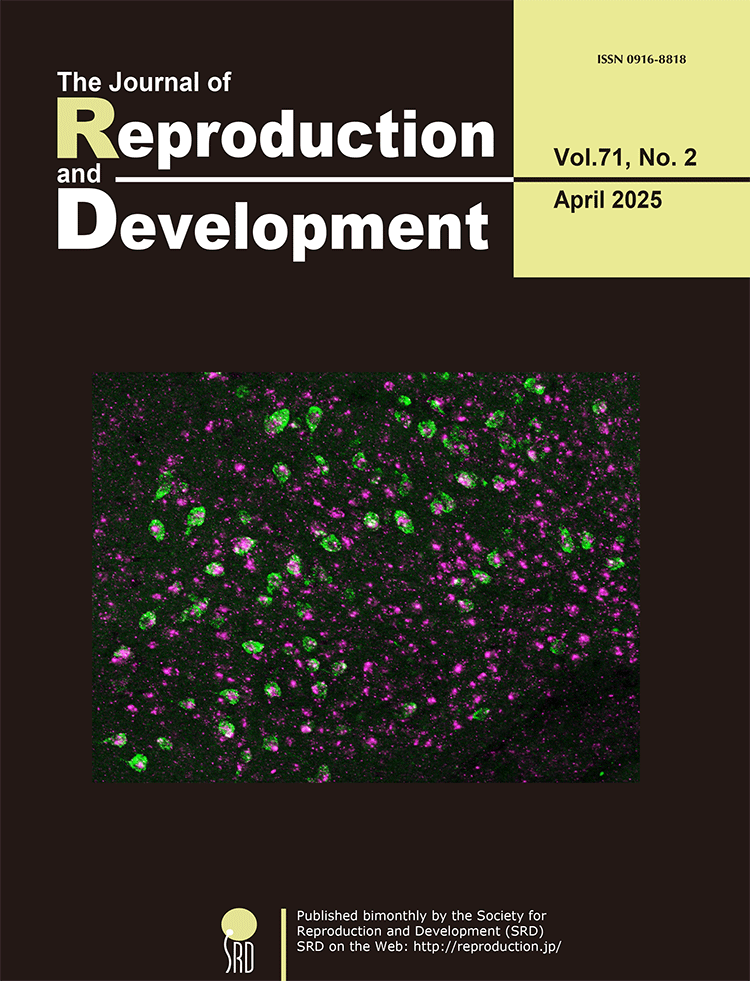
The kisspeptin neurons in the arcuate nucleus (ARC) are the site of estrogen-negative feedback of kisspeptin gene (Kiss1) expression in female mammals. Takizawa et al. investigated whether nuclear receptor corepressor 2 (NCOR2), an estrogen receptor α corepressor, is involved in estrogen-induced Kiss1 repression using two rat models: proestrous virgin and late-lactating model rats (Takizawa et al.; Involvement of nuclear receptor corepressor 2 (NCOR2) in estrogen-induced repression of arcuate Kiss1 expression in female rats. pp. 71–84). Ncor2 (magenta) was expressed in more than 80% of ARC Kiss1-expressing cells (green) in female rats, as shown in the cover photograph. Kisspeptin-neuron-specific Ncor2 knockdown increased the number of Kiss1-expressing cells and the intensity of the Kiss1 signals in the ARC in the proestrous model Kiss1-Cre rats but not in the late-lactating Kiss1-Cre rats. These findings suggest that NCOR2 in ARC kisspeptin neurons mediates the proestrous levels of estrogen-induced repression of ARC Kiss1 expression in virgin rats.
Vol.71,No.1 Cover
The expansion of humanity into space is inevitable. However, human reproduction within space habitats or on extraterrestrial planets poses profound challenges including harmful mutations caused by cosmic radiation and abnormal development of embryos and fetuses in non-terrestrial gravitational environments. Moreover, colonizing other star systems necessitates the transportation of thousands of individuals from each animal species to the target planet to prevent inbreeding-related degeneration. Looking further ahead, as humans disperse throughout the galaxy, the imperative to preserve all genetic resources from Earth permanently and securely becomes paramount. This review examines the issues that must be addressed to ensure human prosperity in space, as well as the challenges that need to be resolved for the transport and long-term preservation of vast genetic resources (Wakayama S. and Wakayama T. Can Humanity Thrive Beyond the Galaxy? pp. 10-16).
Vol.70,No.6 Cover
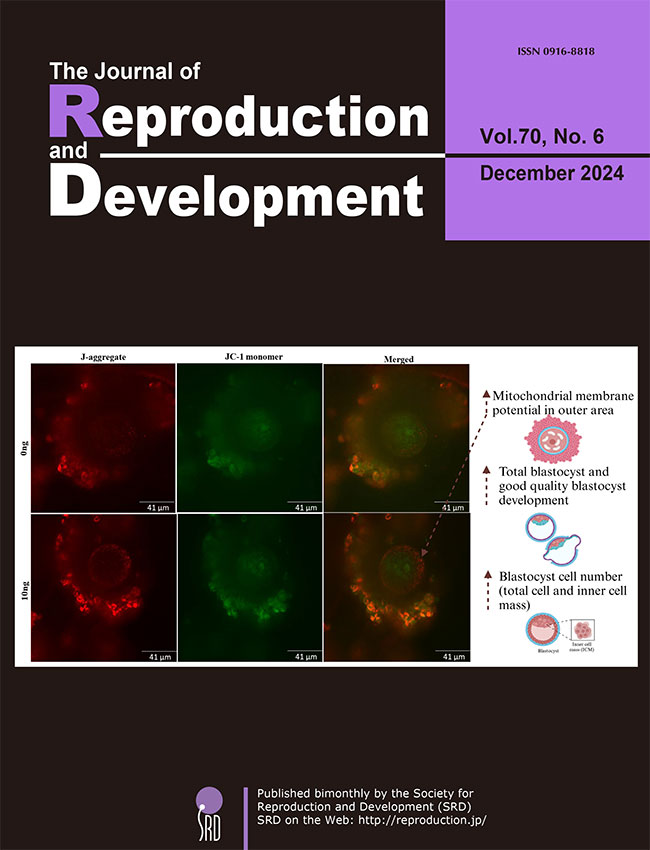
Study by Saini et al. investigated the effects of adding Granulocyte-macrophage colony-stimulating factor (GM-CSF) during in vitro maturation (IVM) on oocyte quality in a mouse model of advanced maternal age (Saini et al.: Addition of granulocyte macrophage colony stimulating factor (GM-CSF) during in vitro oocyte maturation improves embryo development in a mouse model of advanced maternal age. pp. 411–417). Oocytes from older female mice were treated with GM-CSF, and several developmental competence measures were assessed. The treatment tended to increase fertilisation rates (76.19 vs. 82.03; P = 0.07) while increasing blastocyst rates 51.10 vs. 61.52; P < 0.01), and the number of good quality of blastocysts (33.31 vs. 44.13; P < 0.05), along with increased inner cell mass and total cell number. GM-CSF also increased mitochondrial membrane potential. However, it did not affect spindle formation or chromosome alignment. These findings indicate that GM-CSF could improve oocyte quality in women of advanced maternal age by improving embryo development and mitochondrial function.
Vol.70,No.5 Cover
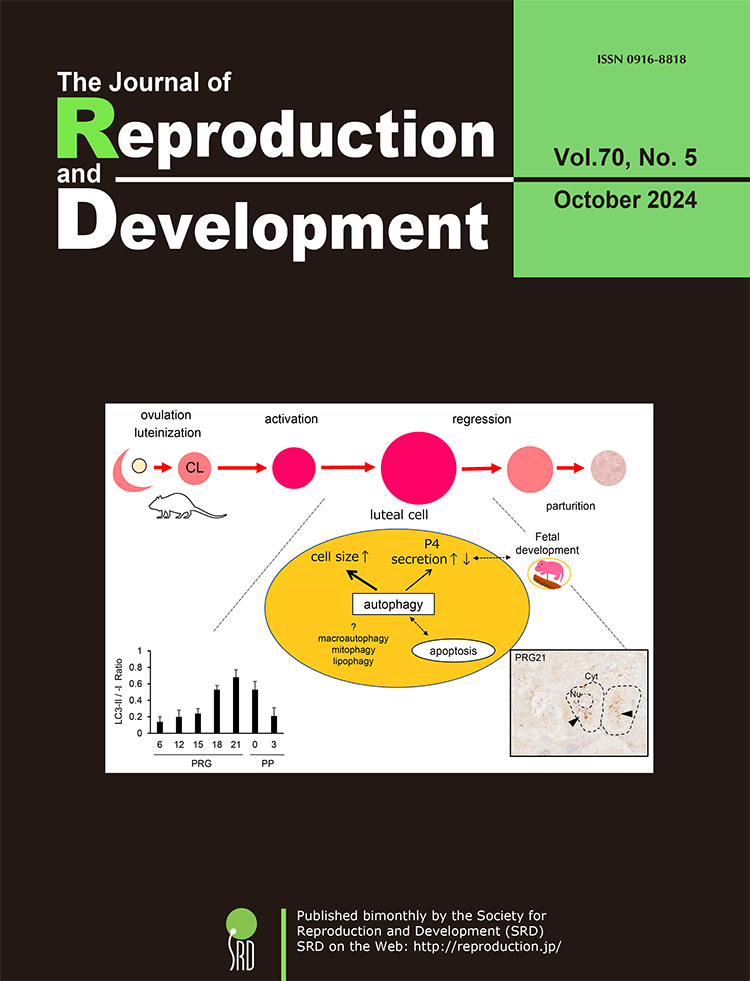
The temporally regulated function and structure of the corpus luteum (CL) are critical for the establishment, maintenance, and termination of pregnancy across various animal species. Oishi et al. found that autophagic activity in the rat CL fluctuates in correlation with tissue weight rather than progesterone (P4) production (Oishi et al., Autophagy in the corpus luteum correlates with tissue growth in pregnant rats. pp. 286–295). Their perturbation experiment using a chemical inhibitor suggests that autophagy contributes to increasing the size of luteal steroidogenic cells and precisely modulates their P4 secretion. The dual nature of autophagy, which controls cellular survival or death, may be implicated in the reciprocal regulation of luteal P4 secretion to determine the appropriate gestational length in species whose P4 production depends solely on CL.
Vol.70,No.4 Cover
Uterine receptivity is the specific state during early pregnancy when the endometrium is ready to accept the embryo or conceptus, and this process by which the embryo attaches to the endometrium is also known as implantation. Kubota summarized the molecular mechanisms underlying steroid hormone-induced uterine receptivity and the similarities and differences in this mechanism among different mammals (Kubota K. Molecular approaches to mammalian uterine receptivity for conceptus implantation, pp. 207–212). This review will help understand the importance of uterine receptivity and the challenges associated with alleviating implantation failure. Investigating the mechanism of uterine receptivity is necessary to develop diagnostic and therapeutic tools for successful pregnancies.
Vol.70,No.3 Cover
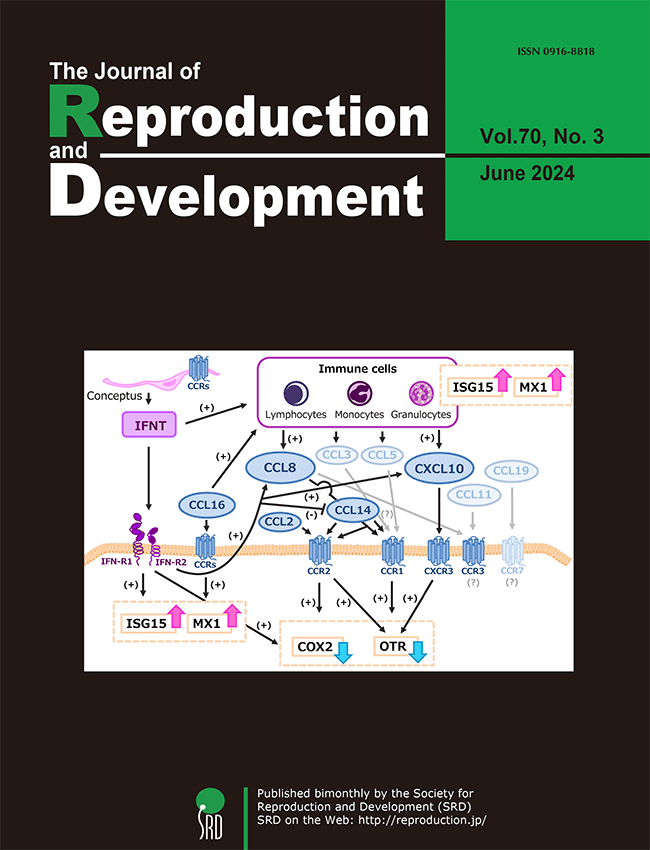
Chemokines are known to regulate various reproductive functions, such as regulation of corpus luteum (CL) and uterine functions in cows. However, the role of chemokines in pregnancy has not yet been fully elucidated. Sakumoto summarized and reviewed the literature on chemokine (-receptor) expression and its physiological roles in the bovine CL and uterus during pregnancy (Sakumoto R. Role of chemokines in regulating luteal and uterine functions in pregnant cows, pp. 145–151). This review will help understand the mechanisms of chemokine-mediated interactions among the CL, uterus, immune cells, and conceptus during pregnancy in cows.
Vol.70,No.2 Cover
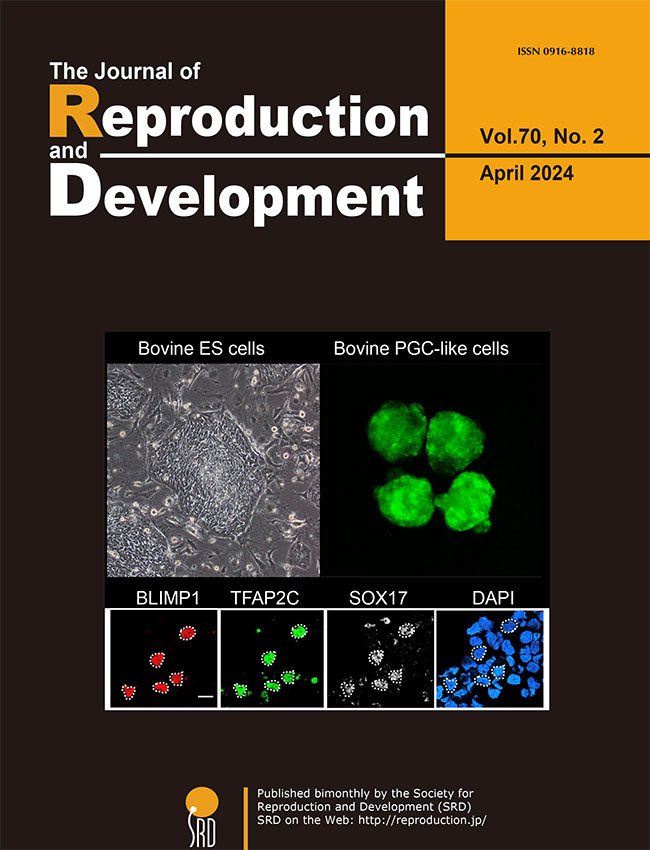
The process of inducing the germ cell lineage from pluripotent stem cells, referred to as
in vitro gametogenesis, aids in comprehending the mechanisms involved in germ cell differentiation and offers an alternative source of gametes for reproduction. Shirasawa
et al. have developed a novel method for robust induction of primordial germ cell-like cells (PGCLCs) from newly established bovine embryonic stem (bES) cells (Shirasawa
et al.: Efficient derivation of embryonic stem cells and primordial germ cell-like cells in cattle, p. 82–95). After a 24-hour culture with bone morphogenetic protein 4 (BMP4), followed by a three-dimensional culture with BMP4 and chemicals modulating WNT signaling, bES cells exhibited positive expression for a set of primordial germ cell (PGC) markers, including PRDM1/BLIMP1, TFAP2C, and SOX17. These outcomes are anticipated to have practical implications for the development of stem cell-based reproductive technologies in cattle.
Vol.70,No.1 Cover
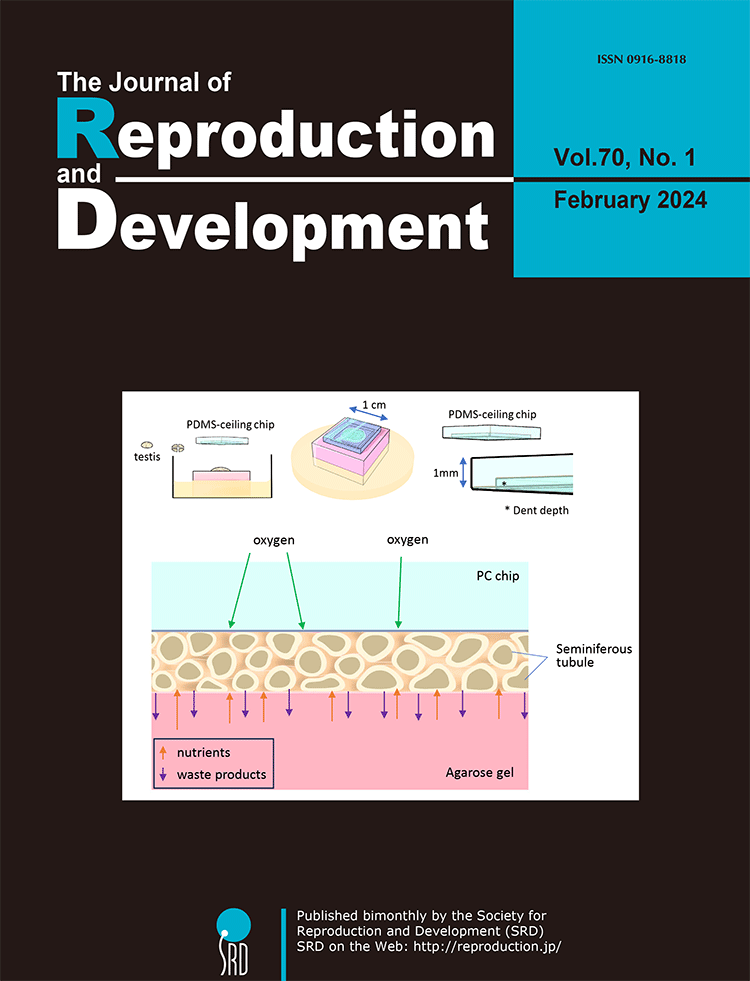
For the past century, achieving in vitro spermatogenesis has remained a difficult challenge for researchers. In 2011, Ogawa et al. successfully demonstrated in vitro spermatogenesis in mice using an organ culture method. However, extending this method to other species posed challenges for over a decade. In 2023, Ogawa’s team achieved in vitro spermatogenesis in rats by incorporating several critical modifications to enhance their original technique. This review presents a detailed analysis by Ogawa et al. comparing their method with natural in vivo conditions and other synthetic alternatives (Ogawa et al. Improvements in in vitro spermatogenesis: oxygen concentration, antioxidants, tissue-form design, and space control, pp. 1–9). They systematically explore the merits, limitations, and inherent constraints of the organ culture approach, delving into the specifics of medium composition, the principles of the gas-liquid interphase method, use of microfluidic devices, and innovation of the PDMS-ceiling method. Highlighting the challenges faced, including regulating oxygen concentration, managing tissue formation, and regulating culture space-control. The insights and novel concepts shared in this review are particularly valuable for those involved in culture or related disciplines, providing innovative content, and encouraging further exploration in this field.
Vol.69,No.6 Cover
Cluster of differentiation 9 (CD9) and sex-determining region Y-box 2 (SOX2) positive cells are stem/progenitor cells for hormone-producing cells in the anterior lobe (AL) of the rat pituitary gland. They are located in the marginal cell layer (MCL) facing the Rathke’s cleft between the AL and intermediate lobe (IL) and the parenchyma of the AL. Horiguchi et al. reported that CD9/SOX2-positive stem cells in the AL-side MCL have potential to supply growth hormone (GH) cells when the increase in GH cell population is required, and the AL parenchyma cells may respond to the demand of thyroid-stimulating hormone (TSH) cell supply in the adult pituitary. (Horiguchi et al. Fluctuation of CD9/SOX2-positive cell population during turnover of GH- and TSH-producing cells in adult anterior pituitary gland, pp. 308–316). These findings may provide one of the mechanisms by which hormone producing cells form from adult stem cells in the pituitary.
Vol.69,No.5 Cover
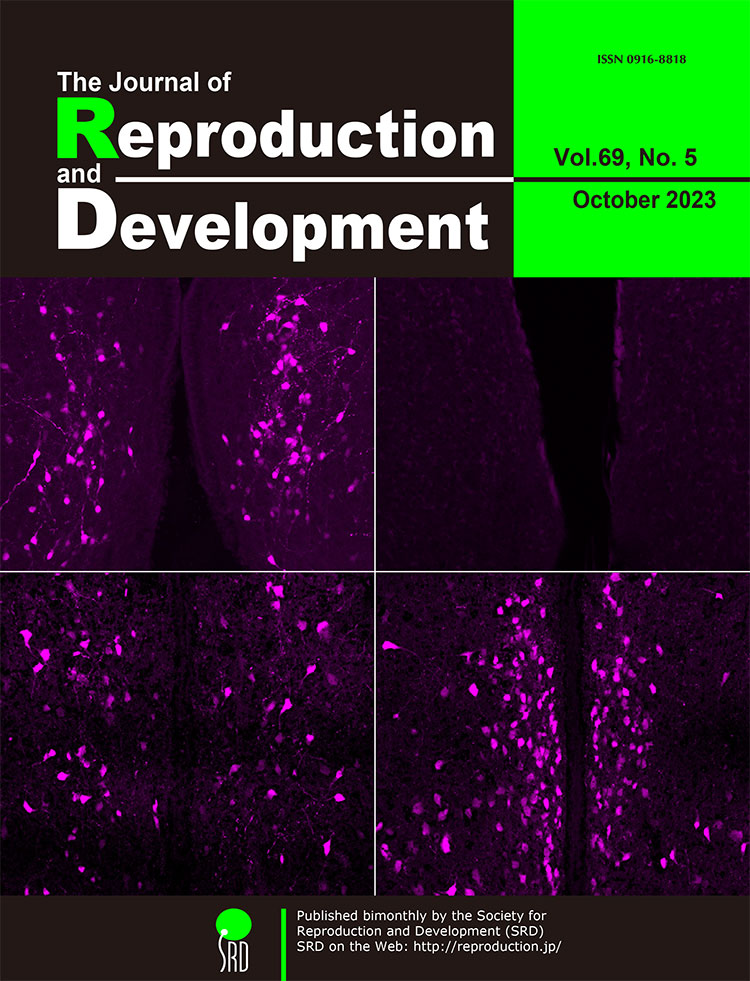
Hypothalamic kisspeptin neurons are master regulators of mammalian reproduction. Yamada et al. generated novel Kiss1 (kisspeptin gene)-Cre rats and investigated the developmental changes and sex differences in visualized Kiss1 neurons of Kiss1-Cre-activated tdTomato reporter rats (Yamada et al.; Sex difference in developmental changes in visualized Kiss1 neurons in newly generated Kiss1-Cre rats, p. 227–238). Histological analysis revealed that Kiss1 neurons, which were visualized by tdTomato, were sexually dimorphic in the anteroventral periventricular nucleus (AVPV), arcuate nucleus, and medial amygdala. As shown on the cover page, neonatal AVPV visualized Kiss1 neurons were detected only in males (upper left; compared to the AVPV in neonatal females on the upper right), but a larger number of visualized Kiss1 neurons were detected in the AVPV in females (lower right) than in males (lower left) in adulthood. The Kiss1-Cre and Kiss1-visualized rats could be valuable tools for further detailed analyses on the sexual differentiation and physiological role of kisspeptin neurons.
Vol.69,No.4 Cover
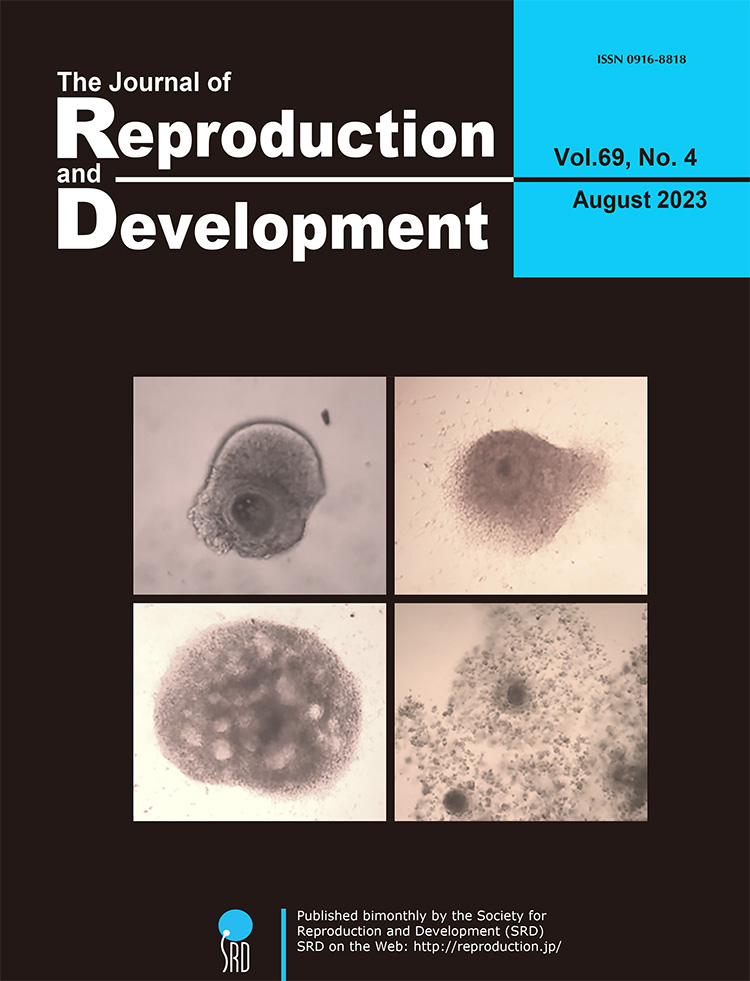
In vitro growth (IVG) culture systems provide a controlled environment for immature and developmentally incompetent oocytes to mature and acquire developmental competence in vitro. However, the reported culture periods have been longer than the follicular development in vivo. To address this, Chelenga et al. developed an 8-day IVG culture system closely resembling the in vivo follicular development period. Notably, this system yielded developmentally competent bovine oocytes when cultured in a gas-permeable culture device. In addition, the combination of the 8-day IVG culture with pre-IVM culture for IVG oocytes significantly enhanced their maturational and developmental competences (Chelenga et al. Pre-maturational culture promotes the developmental competence of bovine oocytes derived from an 8-day in vitro growth culture system, pp. 214–217). The oocyte-cumulus-granulosa complex at Days 0, 4, and 8 of IVG culture (upper left, upper right, and bottom left, respectively) and the matured IVG oocyte after pre-IVM followed by IVM cultures (bottom right) were shown on the cover page.
Vol.69,No.3 Cover

The development of germ cells is accompanied by alterations in the cell cycle in response to external signals and intrinsic cellular mechanisms. During fetal development, male germ cells undergo G0/G1 arrest, whereas female germ cells exit the mitotic phase of the cell cycle and enter meiosis. The NANOS2 and CYP26B1 proteins in the fetal testes cause the germ cells to remain in G0/G1 arrest, which prevents them from entering the meiotic cell cycle. External signals such as RA, BMP, and WNT promote the female germ cells in the fetal ovaries to enter the meiotic phase of the cell cycle. MEIOSIN and STRA8 are transiently co-expressed in the pre-leptotene phase in spermatocytes and oocytes. The MEIOSIN-STRA8 complex ensures the establishment of the meiotic phase by activating meiotic genes in such a manner that the entry into meiosis coincides with the S phase of the cell cycle. This review discusses the development of germ cells from the viewpoint of cell cycle regulation and highlights the mechanism by which germ cells enter the meiotic phase of the cell cycle (Shimada and Ishiguro. Cell cycle regulation for meiosis in mammalian germ cells, pp. 139–146).
Vol.69,No.2 Cover
In chickens, cryopreservation of primordial germ cells (PGCs), the embryonic precursors of gametes, is the best way to cryobank chicken germplasm. However, as cryoprotectants are yet to be optimized for chicken PGCs, the efficacy of cryomedia can be further improved. Hamai et al. designed dimethyl sulfoxide-based and propylene glycol-based cryomedia in conjunction with trehalose and serum that achieved >50% recovery of viable PGCs after thawing while maintaining germline competency (Hamai et al. Development of cryopreservation media for the slow-freezing of cultured primordial germ cells in chicken, pp. 109–117). The offspring of Kurokashiwa, a rare chicken breed in Japan, was successfully revived from PGCs cryopreserved in this cryomedia.
Vol.69,No.1 Cover
Nik-related protein kinase (Nrk) is an X-linked gene encoding a serine/threonine kinase belonging to GCK group 4. Nrk-knockout (Nrk-KO) mice exhibit delayed delivery. However, the mechanism of delayed labor remains largely unknown. Yomogita et al. found that serum progesterone (P4) and placental lactogen (PL-2) concentrations during late pregnancy were higher in pregnant females with Nrk-KO conceptus than in wildtype females (Yomogita et al. A possible function of Nik-related kinase in labyrinth layer of mouse placentas of delayed delivery. pp 32–40). These findings suggest that Nrk-KO mice exhibit delayed delivery due to the increase in P4 concentrations because of PL-2 hypersecretion. Moreover, Nrk was expressed in trophoblast giant cells and syncytiotrophoblast-2 (SynT-2) in the labyrinth layer of the mouse placenta. In the human placenta, NRK is expressed in Syn-T of villi. Similar to mouse Nrk, human NRK may significantly affect placentation in evolutionary biology.
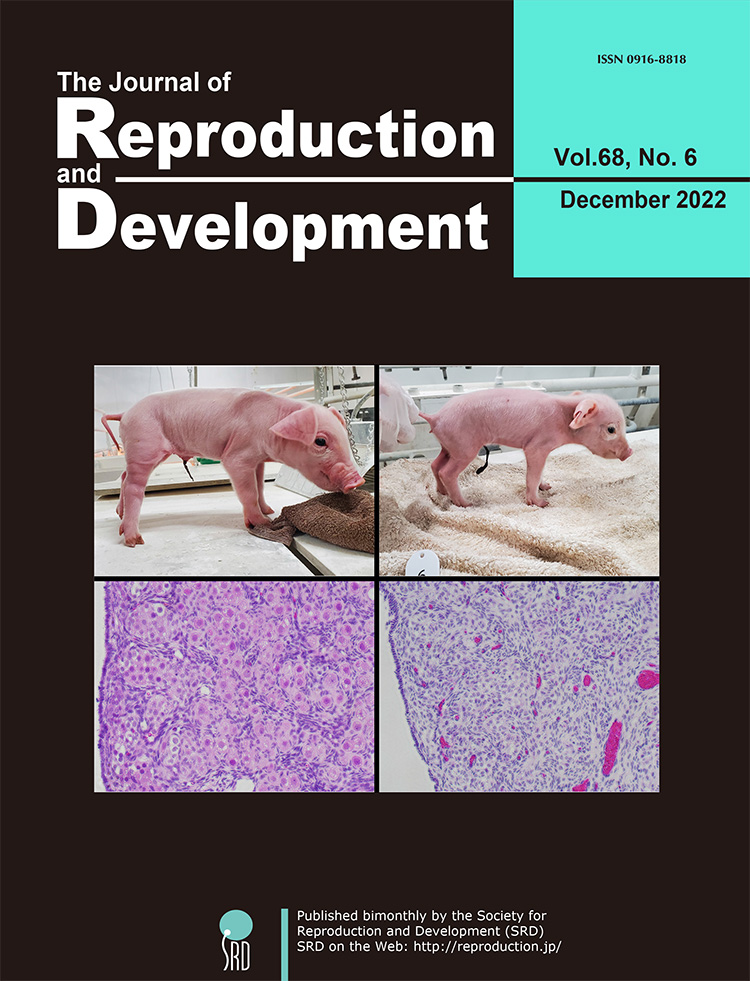
Vol.68,No.6 Cover
NANOS3 is an evolutionarily conserved gene expressed in primordial germ cells, and it is important for germ cell development. Germ cell deletion through NANOS3 knockout has been reported in several mammalian species, although its function in pigs remains unclear. Kogasaka et al. investigated the germline effects of NANOS3 knockout in pigs (Kogasaka et al. Generation of germ cell-deficient pigs by NANOS3 knockout. p. 361–368). Transfer of gene-edited embryos using multiple gRNAs with the Cas9 protein efficiently produced NANOS3-knockout pigs. Histological analysis revealed the lack of germ cells in the testes and ovaries of all NANOS3-knockout pigs. These results demonstrated that NANOS3 is crucial for germ cell production in pigs.
Vol.68,No.5 Cover

The on-gel culture system enables bovine blastocysts to complete cell segregation events at day (D) 10 following in vitro culture [Left panel: Epiblast marker SOX2 (Yellow), Primitive endoderm marker SOX17 (Magenta), DNA marker Hoechst (Cyan)]. This study demonstrated full-term development of D10 mature blastocysts produced using an on-gel culture system (Saito et al. Generation of viable calves derived from developmentally mature blastocysts produced by on-gel culture, pp. 330–334). This confirms that the on-gel culture system recapitulates preimplantation development to D10 and that the in vitro production of D10 blastocysts for cattle production is feasible. Two calves derived from on-gel–cultured embryos were vaginally born. Birth and placental weight were normal for both calves, and no obvious morphological abnormalities were evident. As of August 30, 2022, the calves are still alive and healthy (Right panel). The ability of the on-gel culture system to recapitulate bovine preimplantation development until D10 could contribute to studies on efficient animal production and preimplantation development in cattle.
Vol.68,No.4 Cover
In vitro culture systems, including two-dimensional (2D) and three-dimensional (3D) culture systems, have been used to investigate various cellular functions. Present study has elucidated the effects of these culture systems on the hormonal response and cellular morphology of bovine uterine glands. The gene expression level and hormonal response of secreted proteins differed among isolated (left panels), 2D cultured (middle panels), and 3D cultured (right panels) bovine uterine glands, which might be attributed to their varied cellular morphologies (Sugino et al., Evaluation of bovine uterine gland functions in 2D and 3D culture system, pp. 254–261). Upper and lower panels show fluorescence and phase-contrast images, respectively (red: ZO-1, green: β-catenin, and blue: DAPI).
Vol.68,No.3 Cover

Ovarian functions, such as follicular development and ovulation, are often suppressed in lactating animals. This may be a strategic adaptation to ensure the survival of lactating mothers by avoiding another pregnancy. The suppression of ovarian functions is assumed to be primarily due to the suckling-induced inhibition of hypothalamic kisspeptin neurons (the master regulators of mammalian reproductive function), followed by the inhibition of gonadotropin-releasing hormone (GnRH) and subsequent gonadotropin release. However, the mechanism mediating this inhibition is not fully understood. Sugimoto et al. demonstrated that central antagonism of somatostatin receptor 2 (SSTR2) increased kisspeptin gene expression levels in the hypothalamus of lactating rats, and that some hypothalamic glutamatergic neurons expressed SSTR2. Additionally, SSTR2 antagonism increased luteinizing hormone (LH) release in lactating rats and central glutamate receptor antagonism reversed this effect. Overall, these results suggest that central somatostatin-SSTR2 signaling, at least partly, mediates the suppression of kisspeptin gene expression and subsequent GnRH/LH release by inhibiting glutamatergic interneurons in lactating rats (Sugimoto et al. Central somatostatin-somatostatin receptor 2 signaling mediates lactational suppression of luteinizing hormone release via the inhibition of glutamatergic interneurons during late lactation in rats. pp. 190–197).
Vol.68,No.2 Cover
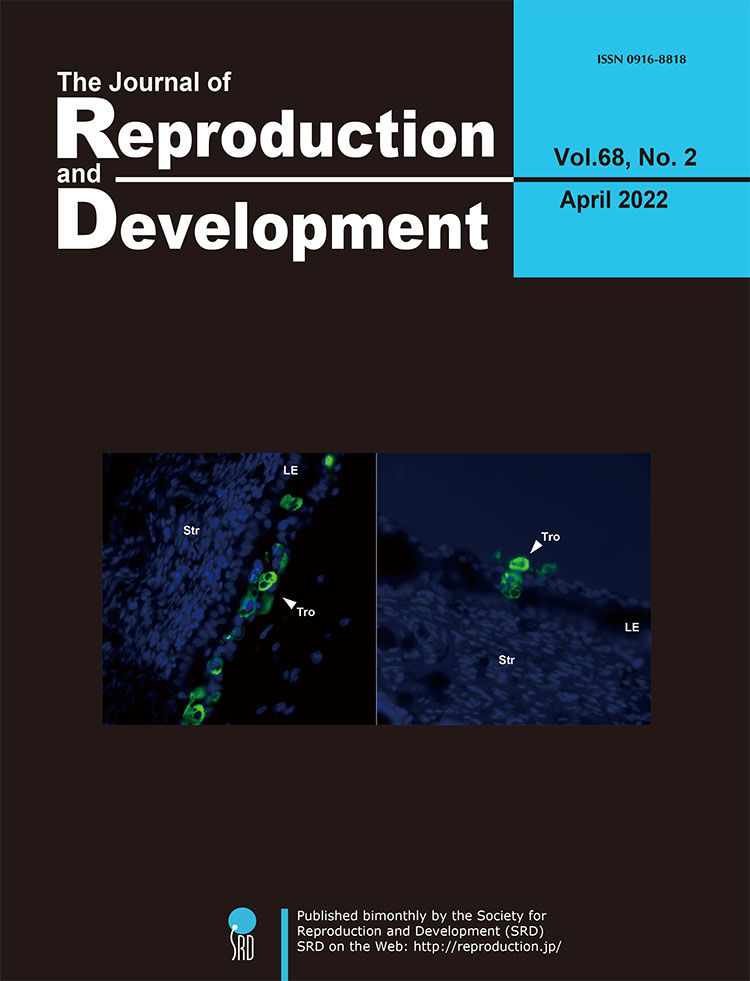
To understand the cellular processes associated with non-invasive mode of conceptus implantation to the maternal endometrium, Yamada et al. observed the conceptus implantation process via adhesion of trophoblast cells to the uterine epithelium (Yamada et al., Epithelial-mesenchymal transition and bi- and multi-nucleated trophoblast cell formation in ovine conceptuses during the peri-implantation period, pp. 110–117). Similar to human syncytiotrophoblast, ruminant trophoblasts form bi- and multi-nucleated trophoblast cells. Using pregnancy associated glycoproteins (PAGs) specific antibody, bi- and multi-nucleated trophoblast cells in cross sections of day 21 ovine uteri (day 0 = day of estrus), including elongated conceptuses, were observed, and it was found that bi- and multi-nucleated trophoblast cells existed on the uterine epithelium or next to the uterine stroma. These results suggest that the formation of bi- and multi-nucleated trophoblast cells facilitates the placental formation in ruminants.
Vol.68,No.1 Cover
Ogata et al. investigated the effects of reduced glutathione (GSH) supplementation in a freezing extender on frozen-thawed semen quality and subsequent in vitro fertilization (IVF) results (Ogata et al. pp. 53–61). GSH supplementation did not affect sperm viability or acrosome integrity (left panel, FITC-PNA staining) in any of the bulls, although few bulls displayed increased DNA damage (right panel, TUNEL staining). However, pronucleus formation and embryonic development after IVF improved in those bulls. These results suggest that supplementing the freezing extender with GSH may improve in vitro embryo production from frozen semen.
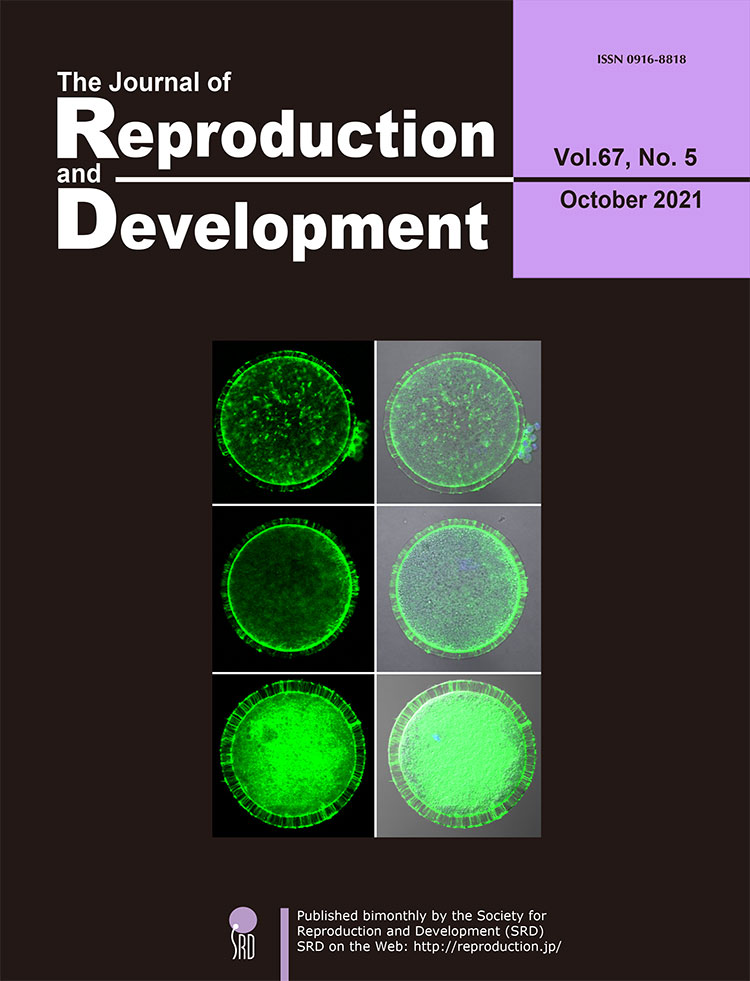
Vol.67,No.6 Cover
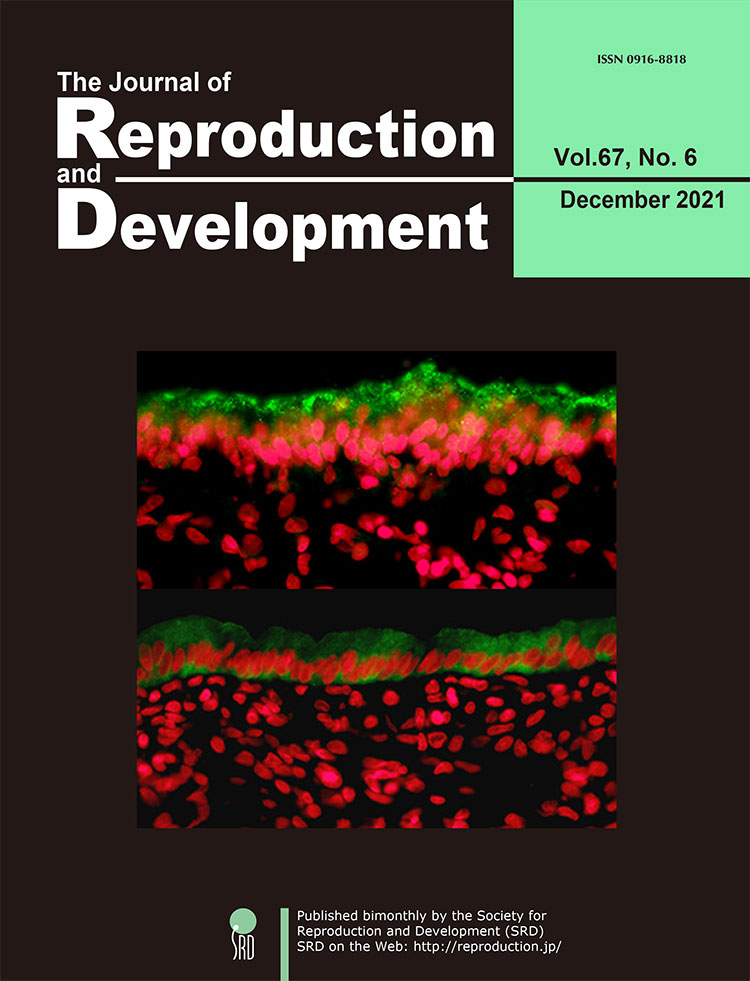
The endometrial surface is required to reject attachment of exogenous undesirable substances such as pathogens, while occasionally shifting properties to allow physical interaction with an embryo to support a successful pregnancy. Kubota et al. reported that MUC1, a potent inhibitor of cellular contact via its large extracellular glycoprotein domain, localizes to the apical surface of bovine endometrium (upper panel, green staining). This MUC1 barrier can be removed in response to steroid signaling at receptive phase and during pregnancy (lower panel), as revealed by immunohistochemistry (p. 386–391). Red signal (pseudo-color) represents DAPI-stained nuclei. Interestingly, MUC1 expression is elevated in the endometrium of cows with a long postpartum interval, which is a known risk for reducing female fertility. Thus, this observation is consistent with the proposed anti-attachment role of MUC1. These results suggest that MUC1 regulation plays a role in endometrial homeostasis and embryo implantation, and its negative correlation with fertility may be used as a diagnostic of reproductive efficiency.
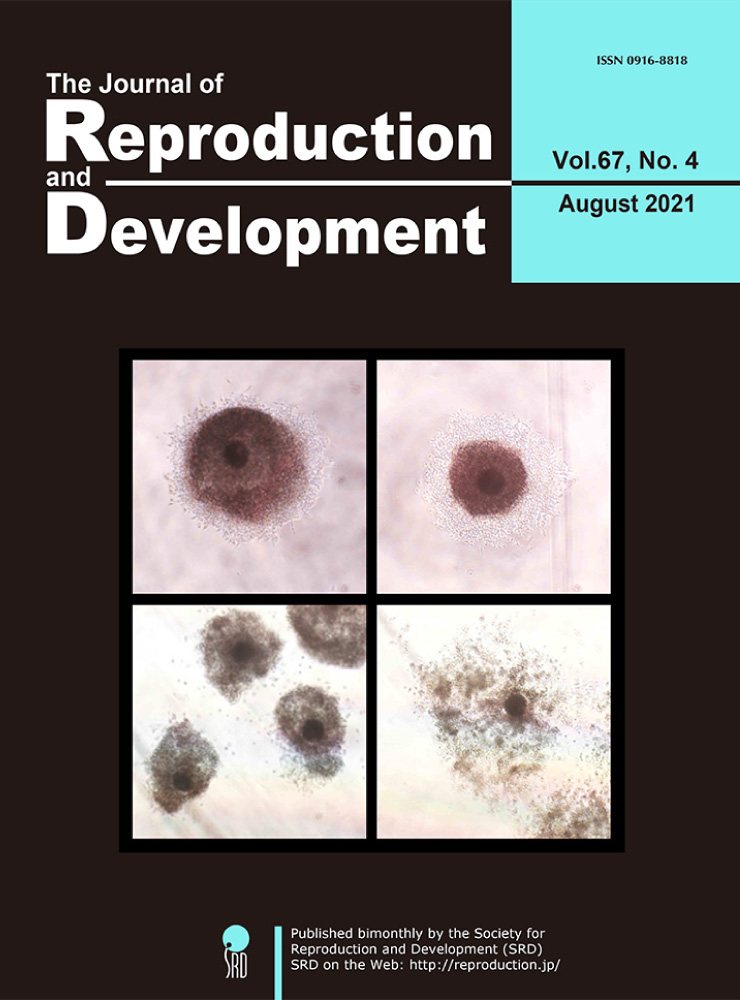
Vol.67,No.5 Cover
Bidirectional communication between oocytes and granulosa cells or cumulus cells is maintained through transzonal projections (TZPs). These structures are important for oocyte growth, although the mechanism underlying TZP development remains unclear. Fushii et al. demonstrated the disappearance of TZPs by denudation of bovine growing oocytes and the reestablishment of TZPs by coculture of TZP-free denuded oocytes (DOs) with mural granulosa cells (MGCs) (Fushii et al. pp. 300–306). Twenty-four hours after denudation, almost all TZPs disappeared, and after coculture and subsequent growth culture of DOs with MGCs, TZPs were reestablished (upper). Additionally, the oocytes in reconstructed complexes grew fully and acquired meiotic competence, suggesting that the reestablished TZPs are able to support oocyte growth similar to those in cultured oocyte-cumulus cell-mural granulosa cell complexes (middle) and in vivo grown oocytes (lower).
Vol.67,No.4 Cover
During oocyte growth and follicle development, oocytes closely communicate with cumulus cells. Morikawa et al. examined the effects of oocyte-derived growth factors, growth differentiation factor 9 (GDF9) and bone morphogenetic protein 15 (BMP15), on the growth and acquisition of meiotic competence of porcine oocytes (Morikawa et al. pp. 273–281). Oocyte-cumulus cell complexes (OCCs) collected from early antral follicles were cultured in growth medium supplemented with GDF9 (upper left) and BMP15 (upper right) for 5 days. Only GDF9 increased the OCC diameter in a dose-dependent manner. OCCs that had been cultured with GDF9 or BMP15 were subjected to a subsequent maturation culture. Those OCCs cultured with GDF9 expanded loosely (lower left), whereas those with BMP15 expanded fully and matured to the second metaphase (lower right).
Vol.67,No.3 Cover
In mammalian female reproduction, primordial follicles serve as stores to sustain the ovulation cycle. Regulations of primordial follicle development, activation, and dormancy in mice are summarized in a review by Nagamatsu (p. 189–195). The importance of mechanical stress, especially extracellular matrix (ECM)-mediated pressure, for the maintenance of primordial follicle dormancy was recently demonstrated. Primordial follicles treated with collagenase, trypsin, and knockout serum replacement (KSR) (this mixture called CTK) to digest ECM were examined by immunohistochemistry. Ovaries treated solely with phosphate-buffered saline displayed primordial follicles composed of flat granulosa cells and complex stress fibers, as revealed by phalloidin. In contrast, CTK treatment resulted in fewer stress fibers and cuboidal-shaped granulosa cells, suggesting oocyte activation.
Vol.67,No.2 Cover
Recent studies suggested that a small sub-population of embryonic stem (ES) cells exhibit 2-cell stage embryo-like (2-cell-like) features, including the reactivation of murine endogenous retrovirus with leucin transfer RNA primer, high histone mobility, and dispersed chromocenters. Furuta et al. investigated the organelle morphology of 2-cell-like cells using electron microscopy (Furuta et al. Lipid droplets are formed in 2-cell-like cells. pp. 79–81). They demonstrated the formation of a lipid droplet during the transition from ES cells to 2-cell-like cells, and proposed that these cells utilize a unique energy storage and production pathway.
Vol.67,No.1 Cover
Previous reports suggested the involvement of alpha-tubulin N-acetyltransferase 1 (ATAT1) in flagella formation in spermatozoa; however, whether ATAT1 is expressed in flagella and involved in spermatozoa maturation and capacitation remains to be elucidated. Yanai et al. evaluated the expression of ATAT1 in the male reproductive system in mice using immunostaining and western blotting (Yanai et al. Expression and localization of alpha-tubulin N-acetyltransferase 1 in the reproductive system of male mice. pp. 59–66). The localization of ATAT1 protein in the male germline was detected during spermiogenesis as well as spermatozoa maturation. Therefore, these results suggest that ATAT1 might be involved not only in flagella formation, but also in the acetylation process during spermatozoa maturation and capacitation
Vol.66,No.6 Cover
Cluster of differentiation 9 (CD9) forms a complex with CD81, and is a member of the tetraspanin superfamily. Cd9 and Cd81 are highly expressed in breast cancer cells, but their expression in healthy mammary glands remains unclear. Horiguchi et al. reported expression of Cd9 and Cd81 in mammary epithelial cells, with expression levels correlating with mammary gland development (Horiguchi et al. Expression and functions of cluster of differentiation 9 and 81 in rat mammary epithelial cells, pp. 515–522). To examine the functional roles of CD9 and CD81, Horiguchi et al. knocked down Cd9 and Cd81 gene expression using small interfering RNAs (siRNAs) in isolated CD9-positive mammary epithelial cells. They found that siRNAs against Cd9 and Cd81 inhibited estrogen-induced mammary epithelial cell proliferation. These findings provide novel insights into mammary epithelial cell proliferation during pregnancy and lactation, as well as in pathological processes associated with breast cancer.
Vol.66,No.5 Cover
Loss of genomic integrity in preimplantation embryos can cause not only developmental arrest but also diseases such as congenital disorders and cancers. However, the key factors for maintaining genomic integrity in preimplantation embryos remain unknown. Shikata et al. reported that SETD8- mediated monomethylation of H4K20, a type of epigenetic modification, plays an important role in the maintenance of genomic integrity (Shikata et al. H4K20 monomethylation inhibition causes loss of genomic integrity in mouse preimplantation embryos. pp. 411–419). In our study, the inhibition of SETD8 or the overexpression of dominant-negative histone H4 mutants resulted in developmental arrest and excessive accumulation of DNA double-strand breaks, suggesting that H4K20 monomethylation is associated with DNA damage repair and is essential for preimplantation development.
Vol.66,No.4 Cover
Spermatogonial stem cells (SSCs) comprise merely a small population in the testis. To facilitate studies on SSCs, it is necessary to find markers that are expressed thereon. Kanatsu-Shinohara et al. discovered a new surface marker on mouse and rat SSCs known as cluster of differentiation 2 (CD2) while studying the molecular mechanism of SSC aging (Kanatsu-Shinohara et al. CD2 is a surface marker for mouse and rat spermatogonial stem cells. pp. 341–349). Using a fluorescence-activated cell sorter, SSCs were enriched by 291.9-fold compared to those in wild-type mouse testes, based on CD2 expression. CD2 depletion by short hairpin RNA in cultured SSCs compromised their stem cell activity. Conserved CD2 expression on mouse and rat SSCs suggest that CD2 may also be expressed on SSCs from other animal species, including humans.
Vol.66,No.3 Cover
To improve the reproductivity of dairy cows, which has been continuously decreasing, it is important to note that the detection of estrus should be accurate and that the timing of artificial insemination (AI) should be appropriately decided. However, the relationship between the conception rate and the timing of AI after the onset of estrus or ovulation period, following the performance of AI using the frozen-thawed semen, has not been investigated yet. However, Sumiyoshi et al. (2020) investigated the relationship between the ovulation time after AI and the conception rate, and identified the time span during which the frozenthawed semen delivers high conception rate after its deposition in the uterus (Sumiyoshi et al., An investigation of the time period within which frozen-thawed semen delivers a high conception rate in lactating dairy cows, pp. 277–280). In this study, the authors demonstrated that high conception rates averaging 63.0% were obtained by performing AI 6–30 h before ovulation, which was significantly higher than the conception rates of 30.0% (P < 0.05) and 26.9% (P < 0.01) obtained from AI that was performed earlier than 30 h before ovulation and later than 6 h before ovulation, respectively. These results further suggest that the frozen-thawed semen can deliver a conception rate of ≥ 60% if it deposits in the uterus for more than 24–30 h after AI, and also that a conception rate of ≥ 60% can be achieved by performing AI 6–30 h before ovulation by using the frozen-thawed semen.
Vol.66,No.2 Cover

Most primordial follicles present in ovaries are dormant and only a few of them are activated in every estrus cycle. However, the mechanism controlling the activation of dormant primordial follicles in vivo remains unclear. In this study, Komatsu et al. found that almost all the activated primordial follicles (black arrows) made contact with blood vessels (red arrows) in mouse ovaries (Komatsu et al. Increased supply from blood vessels promotes the activation of dormant primordial follicles in mouse ovaries. pp. 105–113). To confirm the hypothesis that angiogenesis is crucial for activation of the dormant primordial follicles in vivo, Komatsu et al. induced angiogenesis using recombinant VEGF. They found that the activation of dormant primordial follicles was promoted by an increase in the number of blood vessels in the ovaries. Furthermore, the number of activated follicles increased in cultured ovarian tissues depending on the serum concentration in the medium. These results confirm that the supply of serum components through new blood vessels formed via angiogenesis is a cue for the activation of dormant primordial follicles in the ovaries.
Vol.66,No.1 Cover
Improving artificial oocyte activation is essential for animal biotechnology, to obtain healthy offspring with a high success rate. Yamamoto et al. investigated whether the equine sperm-specific phospholipase C zeta (ePLCζ) mRNA, which has the strongest oocyte activation potential in mammals, could improve the mouse oocyte activation rate and subsequent embryonic development using inactivated spermatozoa (Yamamoto et al. Production of mouse offspring from inactivated spermatozoa using horse PLCζ mRNA. pp. 67–73). The activation potential of ePLCζ was ten times greater than that of murine (m) PLCζ and normal blastocysts were obtained. However, the birth rate was slightly, but significantly, decreased in oocytes activated by ePLCζ compared to those activated by mPLCζ. These results suggest that activation rate does not always correlate birth rate.
Vol.65,No.6 Cover

K Takeda. Functional consequences of mitochondrial mismatch in reconstituted embryos and offspring, pp. 485–489
Animal cloning technology has been developed to produce progeny genetically identical to a given donor cell. However, recipient oocytes contribute a heritable mitochondrial genomic (mtDNA) background to the progeny. In the somatic cell nuclear transfer (SCNT) process, a small amount of donor cellderived mitochondrial genome (D-mtDNA) can accompany the transferred nucleus; therefore, mtDNA from two sources is combined in the cytoplasm (heteroplasmy) of the reconstituted oocyte. The proportion of D-mtDNA can be restricted in animals produced by SCNT due to random distribution, selective replication/degradation associated with cell division during early embryo development, or maternal inheritance of germplasm. These phenomena are described as bottleneck effects (BN1-3). The mtDNA bottleneck characterized by rapid generational shifts in maternal germline can result in both mtDNA heteroplasmic and homoplasmic lines in subsequent generations.
Vol.65,No.5 Cover
The oocyte is the only cell that can reprogram a somatic nucleus to totipotency. The process of reprogramming is, however, only partially understood, and is accompanied by both epigenetic and structural changes in the somatic nucleus. The oocyte components that are necessary for a successful reprogramming and remodeling are unknown. In this issue, Fulka H et al. demonstrate that rather than the insoluble nuclear envelope, together with chromatin-bound factors, or the cytoplasm alone, it is the soluble nuclear fraction that has a major effect upon the somatic nucleus (Fulka H, et al.: Dissecting the role of the germinal vesicle nuclear envelope and soluble content in the process of somatic cell remodeling and reprogramming. pp. 433–441). This fraction is essential for altering the size of the somatic nucleus as well as transcriptional silencing and efficient histone H3.3 incorporation.
Vol.65,No.4 Cover
In vitro/ex vivo egg production has been widely studied in various mammalian species over half of the century to utilize the majority of the immature oocytes stocked in the female ovaries. Recently, the first successful protocol of in vitro oogenesis from primordial germ cells (PGC) has been established by Morohaku K, et al., resulting in the live birth of offspring in mice. The protocol consists of two vital steps; 1) ex vivo organ culture of mouse PGC ovaries to complete the process of follicle formation, with successful incorporation of antagonists for the existing estrogen receptors, and 2) in vitro follicle culture of the growing follicles isolated from the cultured ovaries. The review in this issue introduces the current findings and aspects governing in vitro oogenesis, with a brief history (Morohaku K. A way for in vitro/ex vivo egg production in mammals. pp. 281–287).
Vol.65,No.3 Cover
In cattle, small oocytes (group VII), included in small follicles, grow with follicular development, and the size and developmental competence of oocytes increase (groups I and II) (Nagano, Acquisition of developmental competence and in vitro growth culture of bovine oocytes, pp. 195–201). Subsequently, only one follicle is selected to develop to a dominant follicle and ovulates, but the other follicles start to degenerate. During the degeneration process, an accumulation of lipid droplets and undulation of the nuclear membrane of germinal vesicle start, and the developmental competence of oocytes also increases (pseudomaturation-like change in group III). However, too many pseudomaturation-like changes impair the developmental competence of oocytes (groups V and VI). If oocytes start to degenerate before pseudomaturation-like changes, the oocytes may become group IV.
Vol.65,No.2 Cover

The mitochondrial sheath is composed of mitochondria that coil tightly around the midpiece of the sperm flagellum. Mitochondria are recruited from the cytoplasm to the flagellum late in spermatogenesis. Recruited mitochondria are initially spherical, but then elongate laterally to become crescent-like in shape. Subsequently, these crescent-like mitochondria elongate continuously to coil tightly around the flagellum. Mitochondrial sheath development in glycerol kinase 2 (Gk2)-disrupted mice, which show abnormal mitochondrial sheath formation, was observed using freeze-fracturing coupled with scanning electron microscopy (Shimada et al., Glycerol kinase 2 is essential for proper arrangement of crescent-like mitochondria to form the mitochondrial sheath during mouse spermatogenesis, pp. 155–162). Gk2-disrupted spermatids show abnormal localization of crescent-like mitochondria, despite initially exhibiting proper alignment of spherical mitochondria around the flagellum. These results indicate that GK2 is essential for proper arrangement of crescent-like mitochondria during mitochondrial sheath formation in mouse spermatogenesis.
Vol.65,No.1 Cover

Mammalian oocyte quality degrades over time after in vitro ovulation. As various oocyte manipulations employed in assisted reproductive technology are time consuming, post-ovulatory aging is a serious problem in reproductive medicine and ova research. Shimoi et al. investigated the effects of post-ovulatory aging on the incidence of chromosomal aneuploidy during meiosis II (MII), with a focus on the expression of functional proteins from the spindle assembly checkpoint (SAC) (Shimoi G et al.: Destabilization of spindle assembly checkpoint causes aneuploidy during meiosis II in murine post-ovulatory aged oocytes. pp. 57–66). This study showed that post-ovulatory oocyte aging inhibits MAD2 localization to the sister kinetochore. Furthermore, oocyte aging prevented cohesin subunits from being appropriately maintained or degraded. These results suggest that destabilization of SAC signaling causes sister chromatid segregation errors in MII oocytes and consequently increases the incidence of aneuploidy in early embryos.
Vol.64,No.6 Cover
The review article by Uenoyama et al. describes the roles of hypothalamic kisspeptin in the central mechanism regulating puberty and subsequent reproductive functions in mammals. The schematic illustration shows a possible mechanism regulating the pubertal augmentation of Kiss1 expression in the arcuate nucleus (ARC) to trigger pulsatile GnRH/ gonadotropin secretion in rodents. The authors suspect that estrogen strongly suppresses ARC Kiss1 expression during the prepubertal period via direct and indirect pathways and that the sensitivity to estrogen negative feedback action on ARC Kiss1 expression decreases during the pubertal transition. The resultant increase in secretion of kisspeptin would trigger GnRH/gonadotropin secretion at pubertal onset (Uenoyama et al. The role of kisspeptin in the mechanism underlying reproductive function in mammals. pp. 469–476).
Vol.64,No.5 Cover

Dry biobanking has many advantages over the current paradigm of storing cryopreserved cells under liquid nitrogen. During drying, however, the cells become damaged. The highly condensed spermatozoa DNA has been shown in many desiccation studies to generally maintain its integrity. Using ram freeze-dried epididymal spermatozoa as a model, Palazzese et al. were the first to evaluate both single- and double-strand DNA breaks (SSBs and DSBs, respectively), showing that drying causes minimal DSBs but extensive SSBs (Palazzese L et al., 2018. DNA fragmentation in epididymal freeze-dried ram spermatozoa impairs embryo development, pp. 393–400). Furthermore, the authors also demonstrated that spermatozoa capable of directing embryo development to the blastocyst stage in vitro originated from rams with the least DNA damage Overall, the impact of sperm DNA damage on embryonic development depends on a balance between the extent of sperm DNA fragmentation, fragmentation type, and the oocyte’s repair capacity.
Vol.64,No.4 Cover
Oog1, an oocyte-specific gene, encodes the protein belonging to the leucine-rich repeat (LRR) superfamily. LRR is a motif involved in protein-protein interactions. Complete knockout of Oog1 is challenging because five copies of the Oog1 gene are present on chromosomes 4 and 12. Honda et al. generated Oog1 RNA interference (RNAi)-transgenic mice to investigate the effects of Oog1 knockdown on gene expression in the oocytes (Honda et al. Oocyte-specific gene Oog1 suppresses the expression of spermatogenesis-specific genes in oocytes, pp. 297–301). The abundance of spermatogenesis-specific transcripts was elevated in the Oog1 knockdown ovaries. In addition, a few abnormal oocytes were observed in 6-month-old Oog1 knockdown mouse ovaries. These findings suggested that OOG1 suppresses the expression of spermatogenesis-specific genes in the oocytes and plays important roles during oogenesis.
Vol.64,No.3 Cover

Based on the results obtained for the studies of in vitro development of cloned embryos, epigenetic modifications have been widely used for cloning farm animals. However, such studies remain few in canids because of the lack of optimal in vitro oocyte maturation, embryo culture, and superovulation system. Kim et al. investigated whether a histone deacetylase inhibitor used in dog to pig interspecies somatic cell nuclear transfer (iSCNT), which improves nuclear reprogramming, could be used in dog cloning (Kim et al.: Suberoylanilide hydroxamic acid during in vitro culture improves development of dog-pig interspecies cloned embryos but not dog cloned embryos. p. 277–282). Porcine oocytes supported reprogramming of nuclei from dog fibroblasts up to early developmental stage of iSCNT embryos, and treating the embryos with suberoylanilide hydroxamic acid (SAHA) increased their developmental competence. However, unfortunately, SAHA treatment for dog to pig iSCNT embryos was not sufficient to improve their in vivo development because three and one clones were successfully produced from the control and SAHA treated groups, respectively.
Vol.64,No.2 Cover
Recently, the effects of macromolecular crowding on cultured cells have gathered increasing interest. Crowded culture medium prepared by the addition of polyvinylpyrrolidone (PVP; Mw 360,000) positively influences the viability of oocytes during in vitro growth. Mizumachi et al. found that crowding affects a wide range of factors, including oocyte viability, complex morphology, and intimate conjunction of oocytes with cumulus/granulosa cells across the zona pellucida (Mizumachi et al. Macromolecular crowded conditions strengthen contacts between mouse oocytes and companion granulosa cells during in vitro growth, pp. 153–160). Confocal laser scanning microscopy indicated a higher number of transzonal processes (TZPs) reaching the oocyte from cumulus cells in 2% PVP medium than in 0% PVP medium.
Vol.64,No.1 Cover
Whether cumulus cells are required for the successful vitrification of mature oocytes in cattle is controversial. Ishii et al. re-evaluated the effects of the presence of multi-layered cumulus cells (MCCs) on the vitrification of mature bovine oocytes (Ishii et al., Embryogenesis of vitrified mature bovine oocytes is improved in the presence of multi-layered cumulus cells. pp. 95–99). In the presence of MCCs, there was no difference between the embryonic development of fresh and vitrified mature bovine oocytes. These results suggest that MCCs protect mature oocytes from freezing, and promote their survival and development after in vitro fertilization. Ishii et al. successfully produced calves using a small number of vitrified mature oocytes with MCCs collected from the ovaries of individual cows post-slaughter.
Vol.63,No.6 Cover
Okuyama et al. reported a transvaginal endoscopy-based technique for conducting ovarian examination in sows in a standing position. Sows were sedated in pig stalls, and their vaginal walls were punctured. Subsequently, a urethroscope was inserted into their abdomen, and an examination was conducted after their ovaries had moved towards the urethroscope camera via rectal palpation (Okuyama MW, et al. A transvaginal endoscopy-based technique for performing ovarian examinations in sows. pp. 617–622). This less invasive procedure without the use of general anesthesia may allow repeated ovarian examinations and increase our understanding of the ovarian dynamics in pigs.
Vol.63,No.5 Cover
Geminin is involved in the regulation of DNA replication and serves as a transcriptional molecular switch that directs cell fate decisions. Yuan et al. investigated the functions of geminin in postnatal, pre-meiotic male germ cells by using the Stra8-Cre/loxP system (Yuan et al. Geminin deletion in pre-meiotic DNA replication stage causes spermatogenesis defect and infertility, pp. 481–488). Geminin-deficient mice exhibited smaller testes compared to the control group, concomitant with few spermatozoa in the cauda epididymis and the severe loss of germ cells. These results suggest that geminin plays important roles in pre-meiotic DNA replication and subsequent spermatogenesis.
Vol.63,No.4 Cover
Gonadotropin-releasing hormone (GnRH), a neuropeptide released from the hypothalamus, has a central role in reproductive function in mammals; however, its role in luteal vascularity has not yet been analyzed. Kanazawa et al. investigated the effects of GnRH agonist treatment on Day 5 (Day 0 = estrus) on luteal blood flow and accuracy of pregnancy prediction in recipient cows (Kanazawa et al., Administration of gonadotropin-releasing hormone agonist on Day 5 increases luteal blood flow and improves pregnancy prediction accuracy on Day 14 in Holstein recipient cows, pp. 389–399). The maximum-diameter luteal blood flow areas in GnRH-treated cows were significantly higher than those in the non-treated cows on Days 7 and 14. In addition, the sensitivity and specificity of pregnancy prediction on Day 14 using the paired blood flow area and time-averaged maximum velocity at the spiral arteries were increased by the GnRH treatment. These results suggest that GnRH treatment on Day 5 increases the luteal blood flow on Days 7 and 14, and improves the accuracy of pregnancy prediction in recipient cows on Day 14.
Vol.63,No.3 Cover

Well-organized mitochondrial functions and dynamics are critical for early embryonic development, but their mechanisms are poorly understood. Ren et al. performed a high-resolution dynamic profiling of mitochondria-related genes (MtGs) to acquire a more detailed understanding of mitochondrial modifications during early development (Ren et al. High-resolution profiles of gene expression and DNA methylation highlight mitochondrial modifications during early embryonic development, pp. 247–261). Their results suggest that the resumption of mitochondrial mass growth is facilitated not only by increased mitochondrial biogenesis and mitochondrial DNA (mtDNA) replication, but also by the appropriate balance between mitochondrial fission and fusion (upper figure). In addition, increased levels of reactive oxygen species (ROS) resulting from enhanced mitochondrial function may be the critical inducer for activating the glutathione (GSH)-based stress response system in early embryos (lower figure).
Vol.63,No.2 Cover

During the process of zygotic gene activation (ZGA), which occurs in the fertilized oocyte, genomic function is regulated by dynamic structural changes in the nucleus. Nakaya et al. developed an easy-to-use chamber device (EASI-FISH chamber) for three-dimensional fluorescence in situ hybridization (3D-FISH) in the embryos and succeeded in visualizing the intra nuclear spatial arrangements in the fertilized eggs (Nakaya M, et al., Visualization of the spatial arrangement of nuclear organization using threedimensional fluorescence in situ hybridization in early mouse embryos: A new “EASI-FISH chamber glass” for mammalian embryos. pp. 167–174). Pericentromeric regions which comprise heterochromatin clusters called chromocenters in somatic cells decondensed during the 1- to 2-cell stages and changed to the chromocenter-like structure thereafter. In the early embryos, the ZGA-related Zscan4 gene loci were located peripherally in the CTs but relocated interiorly in somatic cells. In addition, the CTs in the early embryos were enlarged and had deformed shapes, which were quite different from those in somatic cells. This convenient and reproducible 3D-FISH technique for mammalian embryos is a valuable tool that will provide insights into the nuclear dynamics during development.
Vol.63,No.1 Cover
Cell-secreted vesicles, such as exosomes, have been recognized as mediators of cell communication. In recent studies, exosomes were shown to be involved in the regulation of ovarian function including in the induction of cumulus expansion. To date, exosome function has not been investigated in pigs. Matsuno et al. reported the existence of exosome-like vesicles in porcine follicular fluid and examined the effects of these vesicles on cumulus expansion in porcine cumulus-oocyte complexes (Matsuno et al., Effects of exosome-like vesicles on cumulus expansion in pigs in vitro, pp. 51–58). The existence of exosome-like vesicles was confirmed by transmission electron microscopic observation (top left: negative stain method image; top right: cryo-TEM method image). The vesicles labeled with fluorescent tags were incorporated into cumulus and mural granulosa cells in vitro (bottom left and right: vesicles shown in green with white arrowhead). The vesicles had no significant effects on the expansion of porcine cumulus-oocyte complexes in vitro. Therefore, although exosome-like vesicles exist in ovarian follicles, they are not efficient in inducing cumulus expansion in pigs in vitro.
Vol.62,No.6 Cover

Meiosis-specific α-kleisin subunits (RAD21L and REC8) play roles in the formation of axial elements, assembly of the synaptonemal complex, recombination of homologous chromosomes, and cohesion of sister chromatids. However, the exact functions of the individual α-kleisin subunits remain to be elucidated. Using 3D-SIM, Rong et al. found that RAD21L and REC8 were localized at the connection sites between lateral elements and transverse filaments of pachynema with RAD21L locating interior to the REC8 sites (Rong et al.: Meiotic cohesin subunits RAD21L and REC8 are positioned at distinct regions between lateral elements and transverse filaments in the synaptonemal complex of mouse spermatocytes. pp. 623−630). Intriguingly, some bridge-like signals of RAD21L, but not REC8, were observed between unsynapsed regions of axial elements of zygonema. Furthermore, the signals of recombination intermediates overlapped with those of RAD21L to a greater degree than with the signals of REC8. These results highlight the different properties of the two meiotic α-kleisin subunits, and support the view that RAD21L is an atypical cohesin that establishes the association between homologous chromosomes rather than sister chromatids.
Vol.62,No.5 Cover

Appeltant et al. reviewed the involvement of cAMP in maturation regulation, the function of oocyte-secreted factors (OSFs), and the interaction of both in the bidirectional regulatory loop between porcine oocytes and their cumulus cells. (Appeltant et al. Porcine oocyte maturation in vitro: role of cAMP and oocyte-secreted factors—A practical approach, pp. 439–449). In vivo, follicle-stimulating hormone induces luteinizing hormone (LH) receptors and epidermal growth factor (EGF) receptors (EGFR). LH can stimulate the LH receptor, while EGFR can be stimulated by amphiregulin, epiregulin, and betacellulin. In vitro, EGF cannot activate the EGFR and extracellular signal regulated kinase (ERK1/2) signaling in cumulus cells of porcine cumulus oocyte complexes derived from small antral follicles (< 4 mm), leading to a low developmental competence of these oocytes. The co-operative action of dibutyryl cAMP sodium salt and OSFs in an amphiregulin-stimulated in vitro maturation system promotes the EGFR/ERK1/2 signaling pathway, consequently improving the developmental potential of the oocytes.
Vol.62,No.4 Cover

The mechanism of bovine sperm functional regulation in vivo is still largely unknown. By using spermatozoa from elite bulls (top left), Umezu et al. have proposed a new regulation model of bovine sperm functions mediated by the neurotensin (NT) ligand-receptor system (Umezu et al.: Exogenous neurotensin modulates sperm function in Japanese Black cattle. pp. 409–414). They confirmed the expression of neurotensin receptor 1 (NTR1) in the bovine sperm neck region (top right) and the secretion of NT in the bovine uterus and oviduct. They also examined the effects of exogenous NT on bovine sperm functions such as hyperactivation (middle right). Exogenous NT enhanced protein tyrosine phosphorylation in bovine sperm and acrosomal disappearance rates in a dose-dependent manner in vitro (middle left). These results suggest that NT acts as a facilitator of sperm capacitation and acrosome reaction in the female reproductive tract in cattle, thus highlighting the importance of NT-mediated signaling to regulate sperm functions (bottom).
Vol.62,No.3 Cover
Data on pituitary transcription factors have demonstrated the regulatory mechanisms of the three pituitary glycoprotein hormone subunit genes (Fshβ. Lhβ, and Cga) constituting two types of gonadotropins. However, the functional differences between the LIM-homeobox transcription factors LHX2 and LHX3 in the regulation of Fshβ and Cga remain unclear. Yoshida et al. reported the DNA binding properties and transcriptional activities of Cga and Fshβ between LHX2 and LHX3 as well as their localization in the pituitary gonadotrope of rat (Yoshida et al., Transcription of Follicle-Stimulating Hormone Subunit Genes is Modulated By Porcine LIM Homeobox Transcription Factors, LHX2 and LHX3. pp. 241–248). In particular, immunohistochemical analysis demonstrated a rare population of LHX2 in contrast to LHX3 in the gonadotrope. The present study showed that LHX2 and LHX3 differentially regulate Fshβ and Cga.
Vol.62,No.2 Cover

Miyahara et al. revealed that the membrane-bound form of chicken stem cell factor (chSCF2) was highly beneficial for the in vitro culture of chicken primordial germ cells (PGCs) and that it exerted its growth-promoting effects by cooperating with fibroblast growth factor 2 (Miyahara et al., Chicken stem cell factor enhances primordial germ cell proliferation cooperatively with fibroblast growth factor 2, pp. 143–149). The cultured PGCs were maintained as large spheres (differential interference image) on a feeder layer stably expressing chSCF2. These cultured PGCs expressed the germ cell marker CVH (green) and the undifferentiated cell marker SSEA-1 (red). The cultured PGCs were isolated from a white donor chicken and then transplanted into a recipient embryo from a black chicken, generating a germline chimeric chicken expected to possess both donor- and recipient-derived sperm. The presence of a white chick among the offspring of this chimeric chicken demonstrates that these cultured PGCs were able to undergo normal gametogenesis. These results suggest that chSCF2 induces hyperproliferation of chicken PGCs while retaining their germline competency.
Vol.62,No.1 Cover
In vitro growth of immature oocytes provides the opportunity to increase gametic resources and to understand the mechanisms underlying oocyte development. Eppig et al. demonstrated that non-growing oocytes in primordial follicles can develop into functional oocytes in vitro; however, no one has been able to reproduce such excellent data in the last 20 years. Morohaku et al. established a novel and robust protocol for producing mature oocytes from non-growing oocytes in vitro (pp. 1–5). The addition of fetal bovine serum and polyvinylpyrrolidone to the medium is the primary difference between the protocols established by Morohaku et al. and that of Eppig et al. The resulting oocytes can develop into fertile offsprings after in vitro fertilization and embryo transfer. The birth rate, by this method, greatly exceeds that of the previous study. Although there is still scope for further improvement, Morohaku’s report will facilitate the in vitro growth of mammalian oocytes for practical applications.
Vol.61,No.6 Cover

Artificial insemination (AI)-subfertile bulls have occasionally been found in the sire candidates of high-performance Japanese Black cattle. Kishida and Sakase et al. reported that it is valid to perform exact examinations of acrosomal conditions of cryopreserved spermatozoa for exclusion of these subfertile bulls from the AI program for Japanese Black cattle (Kishida and Sakase et al. Effects of acrosomal conditions of frozen-thawed spermatozoa on the results of artificial insemination in Japanese Black cattle, pp. 519–524). The acrosomal conditions were assessed by peanut agglutinin-lectin staining and immunostaining of acrosomal tyrosine-phosphorylated proteins. The percentages of cryopreserved spermatozoa with normal acrosomal conditions were significantly correlated with the conception rates of routine AI, rates of transferable embryos in superovulation/AI-embryo collection tests and in vitro fertilization rates. These results indicate that low conception rates in AI using cryopreserved spermatozoa with poor acrosomal conditions result from reproductive dysfunctions in the processes between sperm insemination into females and early embryo development, probably from failed fertilization of oocytes with cryopreserved spermatozoa.
Vol.61,No.5 Cover
The dynamics of sex chromosomes during spermatogenesis have been unclear in vivo. Otaka et al. showed the localization of sex chromosomes before and after meiosis in mouse testis sections by fluorescence in situ hybridization (FISH) using specific probes for the X and Y chromosomes (Otaka et al. Distribution of the sex chromosome during mouse spermatogenesis in testis tissue sections. pp. 375–381). The positions of the X chromosomes (red) and Y chromosomes (green) were detected in spermatogenic cells (nucleus; blue), and the dynamics of sex chromosomes during spermatogenesis were demonstrated.
Vol.61,No.4 Cover
Although Rb1 is reportedly essential for self-renewal of spermatogonial stem cells (SSCs), its mechanism has remained unknown. Tanaka et al. examined the phenotype of Rb1-deficient SSCs in detail using cultured SSCs and conditional knock-out mice and found that Rb1 is critical for protection from genomic damage, which induces cell cycle arrest and apoptosis (Tanaka et al. The CDKN1B-RB1-E2F1 pathway protects mouse spermatogonial stem cells from genomic damage. pp.305–316). This picture shows increased expression of γH2AX (a double-strand break marker, red) in GFRA1(green)-expressing undifferentiated spermatogonia in 10-day-old Ddx4-Cre Rb1flox/– mice, in which Rb1 is specifically inactivated in germ cells (blue: Hoechst 33342).
Vol.61,No.3 Cover
Monomeric Plum, a far-red fluorescent protein with photostability and photopermeability, is a potentially suitable cell marker for a variety of animal experiments. Watanabe et al. created a transgenic cloned pig systemically expressing Plum (Watanabe et al. Production of transgenic cloned pigs expressing the far-red fluorescent protein monomeric Plum. pp. 169–177). Expression of Plum commenced at the early embryonic stage and was confirmed in all of the tissues/organs examined, including blood cells. Cells expressing Plum could be clearly distinguished in culture or by flow cytometry from those expressing other fluorescent proteins with shorter wavelengths, such as green fluorescent protein and Kusabira-Orange. These results suggested that the cells, tissues, and organs of pigs expressing Plum could be a useful tool in biomedical research.
Vol.61,No.2 Cover
Rejuvenation research provides the latest information on the molecular and cellular mechanisms necessary for most effective therapeutic approaches for human welfare. Collagen is known to be one of the elements necessary for rejuvenation. Chung et al. investigated whether treatment with collagen complexes had beneficial effects on the rejuvenation or reprogramming of adult mouse-derived fibroblasts (Chang et al. Collagen complexes increase the efficiency of iPS cells generated using fibroblasts from adult mice. pp. 145–153). They found that adult-derived fibroblasts cultured with collagen complexes showed a more youthful state, expanded at a higher rate, and exhibited reduced spontaneous cell death than those cultured without collagen complexes. Further, the efficiency of reprogramming of fibroblasts to become induced pluripotent stem (iPS) cells was significantly higher in adult-derived fibroblasts cultured with collagen complexes than in adult-derived fibroblasts cultured alone. As shown in the cover photo, these iPS cells derived from fibroblasts cultured with collagen complexes showed positive staining for stemness markers such as c-myc, Nanog, Oct4, Sox3, and SSEA-1.
Vol.61,No.1 Cover
It is important to identify which pluripotent stem cells (PSCs) are suitable for human medical application; however, it is unclear how the qualitative differences between different types of PSCs should be evaluated. To provide a human ES/iPS cell model, Honsho et al. converted rabbit ES/iPS cells into a naïve-like state and differentiated them in vitro into a neural lineage cells—oligodendrocytes (Honsho et al. Naïve-like conversion enhances the difference in innate in vitro differentiation capacity between rabbit ES cells and iPS cells, pp. 13–19). It is thought that naïve PSCs have much better differentiating ability than primed PSCs. Naïve-like converted ESCs differentiated much more effectively than primed-state ESCs and naïve-like iPSCs. Also, this study demonstrated that naïve-like conversion can increase the slight qualitative differences of primed-state PSCs and can be used as a valuable strategy for examining the innate capacity of human PSCs for therapeutic use.
Vol.60,No.6 Cover
Forkhead box a2 (Foxa2) has been reported to play an important role in uterine function. However, the regulation of Foxa2 expression in the uterus during early pregnancy is still poorly understood. Yamagami et al. investigated the expression and regulation of Foxa2 in the rat uterus in the event of implantation (Yamagami et al. Expression and regulation of Foxa2 in the rat uterus during early pregnancy. pp. 468–475). It was observed that the expression of Foxa2 mRNA was transiently increased at 3.5 days post coitus and downregulated by 17β-estradiol. FOXA2 protein was localized especially in the nucleus of the glandular epithelial cells. Treatment of the epithelial cells with the recombinant Hedgehog protein significantly increased the expression of Foxa2 in vitro. In a nutshell, the expression profile and further localization of Foxa2 suggested that it could play an important role just before implantation and that it may be regulated by E2 and Hedgehog protein.
Vol.60,No.5 Cover
Histone modifications have important roles for biological processes. Therefore, researchers have investigated multiple cells and tissues. However, little is known about histone modification states during spermatogenesis. Shirakata et al. used immunohistochemical techniques to analyze the histone H4 modification pattern during murine spermatogenesis (Shirakata et al. Histone H4 Modification During Mouse Spermatogenesis. pp. 383–387). All the histone H4 modifications were high during the meiotic prophase, and histone H4 acetylation increased around step 10 spermatids. These results provide further insight into the specific relationship between histone H4 modifications and gene expression.
Vol.60,No.4 Cover
The CRISPR/Cas9 system is expected to be a powerful research tool for studies of genome editing in mammals. Fujii et al. verified the efficiency of the CRISPR/Cas9 system in generating triple-knockout mice (Fujii W, et al. One-step Generation of Phenotype-expressing Triple-knockout Mice with Heritable Mutated Alleles by the CRISPR/Cas9 System. pp. 324–327). A substantial proportion of the pups obtained carried loss-of-function mutations in all of the target alleles, exhibiting typical knockout phenotypes or no protein expression from the target genes. The induced mutations were inherited in the next generation, and therefore, the CRISPR/Cas9 system was demonstrated to be an efficient strategy for generating multiple gene knockout animals.
Vol.60,No.3 Cover
Zygote-specific proteasome chaperone (ZPAC), which is specifically expressed in the mouse gonad and zygote, mediates a unique proteasome assembly pathway in the zygote, but the expression profile and function of ZPAC in the testis are not fully understood. Shimizu et al. investigated the possible role of ZPAC during mouse spermatogenesis (Shimizu et al. Possible Role of ZPAC, Zygote-specific Proteasome Assembly Chaperone, during Spermatogenesis in the Mouse, pp. 179–186). In elongating spermatids, ZPAC was expressed until step 10. In addition, intense ZPAC staining was co-localized with staining of annexin V, an early indicator of apoptosis in mammalian cells, in germ cells of the cryptorchid testis, but ZPAC was also expressed in germ cells showing no detectable expression of annexin V. These results suggest that ZPAC plays a role during spermatogenesis.
Vol.60,No.2 Cover

In many seasonal breeding males, inactive spermatogenesis along with low testosterone concentrations has been detected in the non-mating season. However, in raccoons (Procyon lotor), which are seasonal breeders with a mating season in the winter, some males produce spermatozoa actively even in the summer non-mating season. Okuyama et al. reported that 3β-hydroxysteroid dehydrogenase expression may play an important role in regulating the seasonality of testosterone production (Okuyama et al. Changes in the Immunolocalization of Steroidogenic Enzymes and the Androgen Receptor in Raccoon (Procyon lotor) Testes in Association with the Seasons and Spermatogenesis, pp. 155–161). Immunohistochemical observation of testicular tissues, which exhibited active spermatogenesis in the summer, suggested that spermatogenesis in the raccoon testis might be maintained by some mechanism that regulates aromatase cytochrome P450 expression in the synthesis of estradiol and androgen receptor expression in the control of reactivity to testosterone.
Vol.60,No.1 Cover
Recent studies suggest that spermatogonial stem cells (SSCs) are not comprised of a pure population of cells, but the mechanism that underlies SSC heterogeneity has remained unknown. Ishii et al. investigated the contribution of cell cycle status to cell phenotype and SSC activity using fluorescent ubiquitination-based cell cycle indicator transgenic mice (Ishii K, et al. Cell-cycle-dependent Colonization of Mouse Spermatogonial Stem Cells After Transplantation into Seminiferous Tubules. pp. 37–48). Cultured spermatogonia enriched for SSCs in the G1 phase are significantly more efficient than those enriched in the S / G2-M phase in recolonizing the seminiferous tubules of adult mice following microinjection. These results suggested that cell cycle status is an important factor in regulating SSC activity.
Vol.59,No.6 Cover
Mitochondria are reported to be critical in in vitro maturation of oocytes and subsequent embryo development after fertilization, but their contribution to fertilization has not been investigated in detail. Viet Linh et al. investigated the contribution of mitochondria to fertilization using reconstructed porcine oocytes fused with ooplasmic fragments (Viet Linh N, et al. Fertilization Ability of Porcine Oocytes Reconstructed from Ooplasmic Fragments Produced and Characterized after Serial Centrifugations. pp. 549–556). Oocytes fused with mitochondria-rich fragments showed a higher incidence of sperm penetration and male pronuclear formation after in vitro fertilization compared with other types of reconstructed oocytes. These results suggested possible contribution of active mitochondria to fertilizability of oocytes.
Vol.59,No.5 Cover

Cohesin is a multi-subunit protein complex that plays an essential role in sister chromatid cohesion in mitosis and meiosis. Lee discussed the role of a new cohesin subunit, RAD21L (Lee: Roles of Cohesin and Condensin in Chromosome Dynamics During Mammalian Meiosis. pp. 431–436). In contrast to another meiosis-specific cohesin subunit that is expressed throughout meiosis, REC8, expression of RAD21L is restricted to prophase I. In mouse spermatocytes, RAD21L and its mitotic counterpart RAD21 are expressed in a mutually exclusive manner during meiosis: RAD21L is localized along the synaptonemal complex until mid pachytene, whereas RAD21 is localized there from then on. Later studies using knockout mice have demonstrated that RAD21L is involved in the formation of the synaptonemal complex as well as the synapsis and recombination of homologous chromosomes. However, the precise function of each cohesin subunit and the mechanism regulating their spatiotemporal expression remain to be elucidated.
Vol.59,No.4 Cover
Hiraga et al. reported that the localization patterns of lipid droplets in the cytoplasm of porcine oocytes were evaluated as a novel marker for in vitro maturation of oocytes with high developmental competence (Hiraga et al.: Selection of In Vitro-Matured Porcine Oocytes Based on Localization Patterns of Lipid Droplets to Evaluate Developmental Competence. p. 405–408). Porcine oocytes were differentially stained with Nile red (red) and Hoechst 33342 (blue) for visualization of lipid droplets and nuclei, respectively. Localization patterns were divided into 2 classes: lipid droplets localized uniformly in the whole cytoplasm (class I) and those that were centrally located (class II). Class II oocytes showed a significantly higher rate of blastocyst development than class I oocytes
Vol.59,No.3 Cover
Ectopic expression of Xist is one of the major causes of inefficient somatic cell nuclear transfer in mice. It is known that prior injection of Xist-specific siRNA could significantly improve the birth rates of male clones. In this issue, Oikawa et al. report application of the same knockdown strategy to female clones (Oikawa et al.: RNAi-mediated Knockdown of Xist Does Not Rescue the Impaired Development of Female Cloned Mouse Embryos. pp. 231–237). RNA-FISH analysis revealed that siRNA treatment successfully repressed Xist RNA at the morula stage in female clones. However, unlike male clones, there was no significant improvement in the birth rates of the siRNA-injected embryos.
Vol.59,No.2 Cover

Campbell et al. discussed the use of insulin in embryo culture medium to improve murine embryo development, epiblast cell number, and the generation of embryonic stem cell (ESC) colonies (Campbell et al.: Epiblast cell number and primary embryonic stem cell colony generation are increased by culture of cleavage stage embryos in insulin. pp. 131–138). Embryonic outgrowths from early blastocysts and day 5 blastocysts had fewer epiblast cells, generated outgrowths at a lower rate, and were less likely to contain an epiblast than blastocysts plated on day 6. When day 6 blastocysts were cultured in insulin these factors were further improved. This demonstrated that culture of embryos in insulin only improves the quality of embryonic outgrowths when embryos are cultured to day 6 and have lineage commitment prior to plating. These results provide evidence for the usefulness of culturing embryos with insulin for increasing the yield of outgrowths with the potential to give rise to an ESC colony.
Vol.59,No.1 Cover
Bai et al. discussed the currently ascribed functions of the GATA family in trophoblasts during the peri-implantation period (Bai et al.: Expression and potential role of GATA factors in trophoblast development. pp. 1–6). In rodents, implantation occurs soon after blastocyst hatching from the zona pellucida, followed by several dramatic events toward placentation within a few days. In contrast, development of the post-hatching conceptus in ruminants uniquely features a prolonged peri-implantation period involving trophoblast elongation. Although the duration of peri-attachment periods and types of implantation vary, processes leading to conceptus implantation into the maternal endometrium including roles of GATA factors are similar in most mammalian species.
Vol.58,No.6 Cover
Matsumoto et al. report the mRNA distribution of the motin family in the mouse uterus (Matsumoto et al.: Differential expression of the motin family in the peri-implantation mouse uterus and their hormonal regulation. pp. 649–653). Motins are differentially expressed in uterine cells during the peri-implantation period. The postimplantation period from days 5 to 8 of gestation is characterized by expression of Amot and Amotl1 in secondary decidual cells and loss of Amotl2 expression. Steroid hormones such as P4 and E2 regulate differential expression of the motin family in the mouse uterus in a spatiotemporal manner.
Vol.58,No.5 Cover
Cai et al. report a lower than normal population of spermatogenic cells in the seminiferous tubules as well as a decreased number of spermatozoa in the lumen of the epididymides in HSV1-TK transgenic rats (Cai et al.: Accumulated HSV1-TK proteins interfere with spermatogenesis through a disruption of the integrity of Sertoli-germ cell junctions, pp. 544–551). In the testicular tissue of these transgenic rats that ectopically express the HSV1-TK gene by an inherent promoter, accumulation of HSV1-TK proteins causes a disorder in spermatogenesis. Microarray and real-time PCR analyses for testis RNAs suggest disruption of the integrity of Sertoli-germ cell junctions.
Vol.58,No.4 Cover
Naruse et al. report a treatment that elevates the cAMP levels of bovine in vitro matured oocytes, thereby improves the oocytes’ developmental competence and enucleation efficiency (Naruse et al.: Milrinone treatment of bovine oocytes during in vitro maturation benefits production of nuclear transfer embryos by improving enucleation rate and developmental competence, pp. 476–483). The improved enucleation rate is associated with the proximity of the metaphase plate location to the first polar body, indicating that normality of the cytoskeletal organization is crucial for successful enucleation. Adequacy of the treated oocytes as the recipient oocytes for nuclear transfer is indicated by the confocal images of the metaphase II spindle and cortical microfilaments, actin and tublin filaments, and chromosomes.
Vol.58,No.3 Cover
Fulka et al. report production of giant nucleoli obtained by enucleolation of mouse oocytes and their subsequent fusion (Fulka et al.: Production of giant mouse oocyte nucleoli and assessment of their protein content. pp. 371–376). The oocyte nucleoli (termed nucleolus precursor bodies) serve as the basis of active embryonic nucleoli; however, their composition is currently unknown. The enucleolation procedure in combination with colloidal gold protein labeling allows estimation of the average protein content of a single oocyte nucleolus. This information is crucial when levels of individual nucleolar proteins are to be compared between somatic cells and oocyte/embryonic nucleoli.
Vol.58,No.2 Cover
Brüssow et al. report Doppler images and spectral mode of the umbilical cord of a porcine fetus at day 36 of pregnancy using a laparoscopic guided ultrasound probe (Brüssow et al.: Laparoscopy guided Doppler ultrasound measurement of fetal blood flow indices during early to mid-gestation in pigs, pp. 243–247). This approach allows recording of blood flow characteristics of almost all fetuses in the crowded porcine uterus. Such recordings are not feasible by transcutaneous or transrectal ultrasound measurement. Based on the recordings, fetal size and heart rate parameters and flow indices as well as indexes of vascular resistance can be obtained. These measurements are applicable to interpretation of fetal development and blood supply, and intrauterine growth retardation (IUGR) in piglets.
Vol.58,No.1 Cover
Granulosa-theca cell tumors (GTCTs) are the most common ovarian tumors in cattle. Kitahara et al. report, for the first time, the blood profile and tissue expression of anti-Müllerian hormone (AMH) in bovine GTCTs in this issue (Kitahara et al.: Anti-Müllerian hormone profiles as a novel biomarker to diagnose granulosa-theca cell tumors in cattle. p. 98–104). In the present cases of GTCTs, the ultrasound images of ovaries showed three different appearances of tumors, i.e., cystic, solid or both. Neoplastic granulosa cells were histologically labeled with AMH antibody. Blood AMH levels were significantly higher in cattle with GTCTs than in normal animals with cystic ovarian disease or superovulation treatment. The blood AMH level, therefore, may be a more useful biomarker to diagnose GTCTs than the traditional diagnostic aids. The JRD Editorial Board expresses its heartfelt condolences for the sudden death of Prof. Shunichi Kamimura, who was a coauthor of the present article and contributed to the journal as an editor for many years.
Vol.57,No.6 Cover
The 3D reconstruction analysis using confocal microscopy reveals functional interconnections among nonhypophysiotropic terminal nerve (TN)-GnRH3 neurons controlling motivation for reproductive behavior (Review article by Abe and Oka in this issue: Mechanisms of Neuromodulation by a Nonhypophysiotropic GnRH System Controlling Motivation of Reproductive Behavior in the Teleost Brain. p.665–674). Magenta shows immunofluorescence for anti-salmon GnRH (sGnRH), and green shows fluorescence for intracellularly injected biocytin. Approximately 10–15 TN-GnRH3 neurons form a cell cluster on both sides of the brain. It is noteworthy that fine somatic processes and varicose fibers containing GnRH peptides surround the somata (white). This confocal photomicrograph demonstrates the morphological basis for functional synchronization among TN-GnRH3 neurons by auto / paracrine release of GnRH.
Vol.57,No.5 Cover
Mercury light irradiation is a useful tool for observing fluorescence. An original article appearing in this issue (Terashita Y, et al.: Effect of fluorescent mercury light irradiation on in vitro and in vivo development of mouse oocytes after parthenogenetic activation or sperm microinjection. p. 564-571) reports that oocytes would be seriously damaged by irradiation with a short wave-length light for even short period. The cover of the present issues hows immunolocalization of Oct3/4 and Cdx2 in the blastocysts derived from oocytes irradiated with mercury light following ICSI. The quality of blastocysts irradiated with 341 nm light for 5 sec (at the bottom) is poorer than that of blastocysts irradiated with 341 nm light for 1 sec (at the top) in terms of cell number and Oct3/4 and Cdx2 expression pattern.
Vol.57,No.4 Cover
Arai et al. (Epigenetic Assessment of Environmental Chemicals Detected in Maternal Peripheral and Cord Blood Samples, p. 507–517) introduces a simple and sensitive method to detect het-erochromatin changes together with DNA methylation analysis to identify epimutagenic chemicals (epimutagens) that may affect early embryonic development. Exposure of mouse embryonic stem (ES) cells to certain chemicals, such as Se, as shown in the cover picture, was revealed to cause epigenetic changes (upper and lower panels: negative control and Se exposure, respectively), which were visualized by DNA-FISH with a specific probe for a heterochromatin marker, major satellite (red), merged with heterochromatin dot signals by DAPI staining (blue). Most importantly, their study has, for the first time, proven five chemicals(DEP, Hg, cotinine, Se and S-421) to be epimutagens even at the low concentrations that were detected in maternal cord blood samples.
Vol.57,No.3 Cover
Bovine oocytes were differentially immunostained by anti-tubulin (green) and DAPI (blue) for visualizing the microtubule network assembly and the position of nuclear materials, respectively. The proportion of oocytes that were injected with freeze-dried (FD) sperm and formed a sperm-aster was slightly lower than that of control ICSI oocytes. Among the oocytes exhibiting sperm-aster formation, the extent of microtubule net-work assembly was comparable between the FD-ICSI and control ICSI groups. However, the microtubule-organizing center of the ICSI oocytes was not as functional as that of IVF oocytes in terms of the aster formation rate and the fluorescent intensity of the microtubule network. (Hara H, et al.: Procedure for bovine ICSI, not sperm freeze-drying, impairs the function of the micro-tubule-organizing center. p.428–432)
Vol.57,No.2 Cover
The review article by Inoue et al. in this issue (Inoue N, et al.: Role of cell-death ligand-receptor system of granulosa cells in selective follicular atresia in the porcine ovary. p. 169–175) describes that the apoptotic cascade via the death ligand/receptor system is activated in the granulosa cell during follicular atresia in the porcine ovary. Acivated caspase-3 (green) was found in granulosa cells with fragmented nuclei (red, PI staining) but not in cumulus cells in porcine progressed atretic follicles. Caspase-3 is an effector caspase that induces apoptotic cell death.
Vol.57,No.1 Cover
The vitrified-warmed bovine secondary follicle grew in size in a bovine plasma-supplemented medium. No distinct cavities were found in the follicle after 1 week of culture (top/left), but small cavities appeared after 2 weeks (top/right), which developed progressively larger after 3 weeks (bottom/left) and 4 weeks (bottom/right), eventually forming an antrum-like structure. The combination of cryopreservation and in vitro culture of growing ovarian follicles may be a promising method of providing a large number of mature oocytes. (Taketsuru, et al.: Bovine oocytes in secondary follicles grow in medium containing bovine plasma after vitrification. p.99–106).
Vol.56,No.6 Cover
Compared to untreated embryo (top/left), nuclei with a concave-like shape was observed in the pronuclei of 1-cell embryos transiently treated with MG132 in the G1 phase of the first cell cycle (top/right, and bottom/right and left), which may be similar to the unique structures termed as nucleolar caps. Interestingly, formation of nucleolar caps is known to be a normal cellular process associated with transcriptional shut down and drug-induced transcriptional arrest. The ubiquitin-proteasome system may be involved in transcriptional regulation in early mouse embryos. (Shin SW, et al.: Inhibition of the ubiquitin-proteasomes ystem leads to delay of the onset of ZGA gene expression. p. 655-663).
Vol.56,No.5 Cover
Most centromeres were localized around the nucleoli in the zygote (CREST in green). In addition, oocyte nucleolus loss alters the localization and size of centromeres in both male and female pronuclei, indicating that the nucleolus in the mouse oocyte organizes both paternal and maternal centromeric heterochromatin in pronuclei. The chromatin was stained positive for trimethylated histone H3 at lysine 9 (in red) in female pronuclei and negative in the male pronuclei. (Ogushi S and Saitou M: The Nucleolusin the Mouse Oocyte is Required for the Early Step of Both Female and Male Pronucleus Organization. p. 495–501)
Vol.56,No.4 Cover
In the high fecundity mouse line FL1 of the Leibniz Institute for Farm Animal Biology (FBN, Dummerstorf), the number of ova ovulated was higher than the number of corpora lutea (CL) present. This observation suggests that individual follicles release more than one oocyte. Twenty-seven polyovular follicles per ovary were observed in this mouse line with some follicles containing up to 7 oocytes per follicle. The example shows a polyovular antral follicle with 3 oocytes in a mouse line selected for high fecundity. (Alm H, et al.: Occurrence of polyovular follicles in mouse lines selected for high fecundity. p. 449–453)
Vol.56,No.3 Cover
The localization and expression of chicken germline-specific proteins cDAZL, CDH and CVH are distinct in germ cells during the latter stage of gametogenesis. CDH (red) is found in the nucleus throughout oogenesis. CVH (green) is found in the nucleus and cytoplasmic compartments, although some oocytes are CVH-negative or have weak staining. However, cDAZL is not found in all oocytes (top/right), although cells positive for cDA-ZL and CVH coexist in the embryonic gonads. The lack of cDAZL may imply the initiation of meiosis. Blue: nuclear staining. (Kito G, et al.: Temporal and spatial differential expressionof chicken germline-specific proteins cDAZL, CDH and CVH during gametogenesis. p. 341–346)
Vol.56,No.2 Cover
Immunolocalization of cellular FLICE-like inhibitory protein (cFLIP) in the bovine corpus luteum (CL) at the mid luteal stage. Hojo et al. (in this issue, pp. 230–235) show intense cFLIP immunoreactivities in luteal cells and endothelial cells that remain high during the early, developing and mid luteal stages and low in the late and regressed luteal stages. In addition, exposure of luteal cells to inflammatory cytokines decreased cFLIP expression and increased the number of apoptotic cells. They conclude that cFLIP plays a survival role in the bovine CL.